Products Categories
| CAS No.: | 3637-31-8 |
|---|---|
| Name: | 2H,2H,3H,3H-Perfluoropentanoic acid |
| Article Data: | 6 |
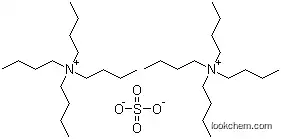
| Molecular Structure: | |
|
|
|
| Formula: | C5H5F5O2 |
| Molecular Weight: | 192.086 |
| Synonyms: | 2H,2H,3H,3H-Perfluoropentanoic acid; |
| Density: | 1.443±0.06 g/cm3(Predicted) |
| Boiling Point: | 158.6±35.0 °C(Predicted) |
| PSA: | 37.30000 |
| LogP: | 2.04880 |
- 483366-12-7(2S,4R)-1-Boc-2-cyano-4-hydroxypyrrolidine
- 5156-58-1N-(1-Benzyl-4-pipperidinyl)-N-phenylpropanamide HCl
- 81281-59-67-Benzylideneaminotheophylline
- 50288-62-5threo-Phenyl-2-piperidyl acetamide
- 82993-81-5D-threo-Ritalinic acid hydrochloride
- 47087-37-6Z-D-Glu-OMe
- 73441-42-6METHYL-5-CHLORO-2,2-DIMETHYLVALERATE
- 68439-39-4Poly(oxy-1,2-ethanediyl), alpha-(2-ethylhexyl)-omega-hydroxy-,
- 852475-26-4MC1568
- 43224-75-52,2,6,6-Tetramethyl-4-(2-propyleneoxy) Piperidine
- 148043-73-6
4,4,5,5,5-pentafluorpentan-1-ol

- 3637-31-8
4,4,5,5,5-pentafluoropentanoic acid

| Conditions | Yield |
|---|---|
| With Jones reagent In acetone at 0 - 20℃; for 1h; | 82% |
| With sodium permanganate monohydrate; tetraethylammonium hydrogen sulphate In water at 65 - 75℃; for 6.5h; Large scale; | 77% |
| With chromium(VI) oxide; acetic acid at 0 - 20℃; for 26h; | 63% |
- 148043-72-5
4,4,5,5,5-pentafluoropentyl acetate

- 3637-31-8
4,4,5,5,5-pentafluoropentanoic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 92 percent / aq. NaOH / 1 h / Heating 2: 63 percent / CrO3, aq. AcOH / 26 h / 0 - 20 °C View Scheme |
- 148043-71-4
4,4,5,5,5-pentafluoro-2-iodopentyl acetate

- 3637-31-8
4,4,5,5,5-pentafluoropentanoic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 78 percent / azoisobutyronitrile (AIBN), Bu3SnH / 55 - 65 °C 2: 92 percent / aq. NaOH / 1 h / Heating 3: 63 percent / CrO3, aq. AcOH / 26 h / 0 - 20 °C View Scheme |
- 32503-27-8
tetra(n-butyl)ammonium hydrogensulfate

- 148043-73-6
4,4,5,5,5-pentafluorpentan-1-ol

A
- 380-60-9
3,3,4,4,4-pentafluorobutanoic acid

B
- 3637-31-8
4,4,5,5,5-pentafluoropentanoic acid

C
- 107-92-6
butyric acid

| Conditions | Yield |
|---|---|
| With potassium permanganate In water at 65 - 70℃; |


| Conditions | Yield |
|---|---|
| With thionyl chloride for 2h; Heating / reflux; | 54% |
- 1333236-67-1
(2,3-dihydrothieno[3,4-b][1,4]oxathiin-3-yl)methanethiol

- 3637-31-8
4,4,5,5,5-pentafluoropentanoic acid

- 1333236-78-4
S-(2,3-dihydrothieno[3,4-b][1,4]oxathiin-3-yl)-methyl 4,4,5,5,5-pentafluoropentanethioate

| Conditions | Yield |
|---|---|
| Stage #1: 4,4,5,5,5-pentafluoropentanoic acid With 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In dichloromethane at 20℃; for 0.5h; Stage #2: (2,3-dihydrothieno[3,4-b][1,4]oxathiin-3-yl)methanethiol In dichloromethane at 50℃; for 24h; | 25% |

| Conditions | Yield |
|---|---|
| With triethylamine In tetrahydrofuran at -78 - 0℃; for 0.333333h; |
- 3637-31-8
4,4,5,5,5-pentafluoropentanoic acid

- 148043-92-9
(-)-(S)-2-azido-4,4,5,5,-pentafluoropentanoic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1: Et3N / tetrahydrofuran / 0.33 h / -78 - 0 °C 2: 1.) BuLi / 1.) THF, hexane, -78 deg C, 2.) THF, hexane, -78 deg C, 20 min 3: 1.) (i-Pr)2NEt, Bu2BOTf, 2.) NBS / 1.) CH2Cl2, from -78 deg C to 0 deg C, 75 min, 2.) CH2Cl2, -78 deg C, 1.25 h 4: tetramethylguanidinium azide / CH2Cl2 / 3 h / 0 °C 5: 70 percent / aq. H2O2, LiOH / tetrahydrofuran / 0.5 h / 0 °C View Scheme |
- 3637-31-8
4,4,5,5,5-pentafluoropentanoic acid

- 148043-77-0
(4R)-3-(4,4,5,5,5-pentafluoro-1-oxopentyl)-4-(phenylmethyl)-2-oxazolidinone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: Et3N / tetrahydrofuran / 0.33 h / -78 - 0 °C 2: 1.) BuLi / 1.) THF, hexane, -78 deg C, 2.) THF, hexane, -78 deg C, 20 min View Scheme |
- 3637-31-8
4,4,5,5,5-pentafluoropentanoic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: Et3N / tetrahydrofuran / 0.33 h / -78 - 0 °C 2: 1.) BuLi / 1.) THF, hexane, -78 deg C, 2.) THF, hexane, -78 deg C, 20 min 3: 1.) (i-Pr)2NEt, Bu2BOTf, 2.) NBS / 1.) CH2Cl2, from -78 deg C to 0 deg C, 75 min, 2.) CH2Cl2, -78 deg C, 1.25 h View Scheme |