Evaluation Only. Created with Aspose.PDF. Copyright 2002-2021 Aspose Pty Ltd.
Ó 2005 The Chemical Society of Japan
Bull. Chem. Soc. Jpn., 78, 1575–1579 (2005) 1575
Heteropoly Blue as a Reaction-Controlled Phase-Transfer
Catalyst for the Epoxidation of Olefins
ꢀ;1;2
Li Mingqiang
and Jian Xigao1
1Department of Polymer Science and Materials, Dalian University of Technology, Dalian 116012, P. R. China
2303group, Dalian Institute of Chemical Physics, Chinese Academy of Sciences, Dalian 116023, P. R. China
Received March 3, 2005; E-mail: limingqiang@dicp.ac.cn
A new reaction-controlled phase-transfer catalyst system has been designed and synthesized. In this system, het-
eropoly blue, [C7H7N(CH3)3]3PMo4O16, is used for the catalytic epoxidation of olefins with H2O2 as the oxidant. In this
system, the catalyst not only can be recovered, like heterogeneous catalyst, but also acts as a homogeneous catalyst. The
main products are epoxide of olefins and H2O; no co-product forms. The system exhibits high conversion and selectivity
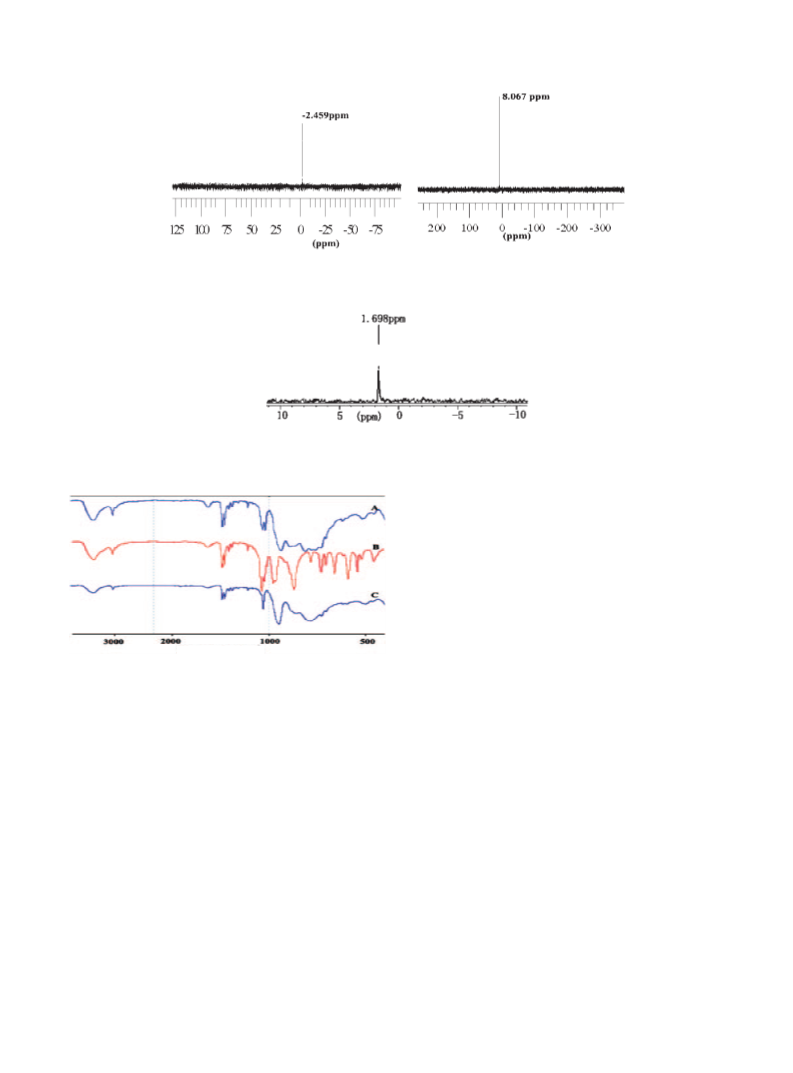
as well as excellent catalyst stability. 31P NMR spectra, UV–vis spectra, and infrared spectra are used to analyze the
reason for the phase transfer of the catalyst, indicating that the change of structure leads to the formation of reac-
tion-controlled phase-transfer catalyst.
Epoxidation—oxygenation of olefins to form cyclic epoxide
groups—is an important industrial process.1–3 Although a
number of epoxidation processes that use various catalysts
and oxidants have been developed, a chlorine-using noncata-
lytic process (the chlorohydrin process) and catalytic processes
based on expensive oxidants are still used extensively.4 In con-
trast to such classical ‘‘nongreen’’ processes, H2O2-based cat-
alytic epoxidation is of great advantage to the environment and
industry because it generates H2O as a sole by-product, and it
is rather inexpensive compared with organic peroxides and
peracids.5 Recently, a reaction-controlled phase-transfer cata-
lyst system has been developed.6,7 In this catalyst system,
the catalyst, which is insoluble in the reaction medium, forms
soluble active species by the action of one of the reactants.
This kind of catalyst not only can be recovered, like a hetero-
geneous catalyst, but also acts as a homogeneous catalyst.
Since the reaction-controlled phase-transfer catalyst system
possesses the above advantages, it is significant to design
and synthesize.
There has been a great deal of interest in the redox behavior
of heteropolyoxometalates and their ability to form ‘‘hetero-
poly blues’’ upon reduction. In the presence of an acid, elec-
trons are usually transferred in even numbers due to stabiliza-
tion of these species by protonation. Numerous heteropoly
complexes can be reduced by the addition of various numbers
of electrons. The reduction products, which typically retain the
general structures of their oxidized parents, and are frequently
deep blue in color, comprise a potentially important large
group of complexes, generally known as heteropoly blues.
The added (‘‘blue’’) electrons are ‘‘delocalized’’, according to
various time scales, over certain atoms or regions of the
heteropoly blues, corresponding to class II systems in the
Robin and Day classification of mixed-valence compounds.
In the present work, we designed and synthesized a reac-
tion-controlled phase-transfer catalyst comprised of heteropoly
blue, [C7H7N(CH3)3]3PMo4O16, for the catalytic epoxidation
olefins with 30% H2O2 as the oxidant. In same time, we inves-
tigated the reason of solid–liquid–solid phase cycle of the
catalyst. The catalyst has high efficiency of H2O2 utilization
and selectivity. Moreover, the reason for the solid–liquid–solid
phase cycle of the catalyst heteropoly blue, [C7H7N(CH3)3]3-
PMo4O16, is different from that of others catalyst.
Experimental
Unless specially mentioned, the chemicals used in this work
were at least of C. P. grade and were used as purchased.
Preparation of Heteropoly Acid [C7H7N(CH3)3]3PMo4O16.
Preparation method according to the literature:8 10 mmol
Na2MoO4 2H2O was dissolved in 30 mL of water. To this solu-
ꢁ
tion was added dropwise 2.5 mmol of 85% H3PO4, and added
with stirring the required amount of ice acetic acid to adjust the
pH to 4. To the resultant solution, 5 mmol of benzyltrimethyl-
ammonium chloride in water (15 mL) was added dropwise with
stirring over about 30 min at 60 ꢂC. The precipitate, being the
product, was filtered, washed with water, and air-dried at room
temperature. The yield was 83%. Anal. Calcd for [C7H7N-
(CH3)3]3PMo4O16: C, 32.09; H, 4.28; N, 3.70; Mo, 34.28; P,
2.79%. Found: C, 32.13; H, 4.31; N, 3.75; Mo, 34.22; P, 2.76%.
IR (neat, KBr plates): 3433, 3034, 1629, 1456, 1217, 1063,
1038, 938, 889, 812, 780, 703, 517 cmꢃ1
Preparation of [C7H7N(CH3)3]3PO4{MoO(O2)2}4.
.
The
[PO4{MoO(O2)2}4]3ꢃ was reported in the literature,9 and convert-
ed to the corresponding tetrabutylammonium salts by a cation-
exchange reaction with benzyltrimethylammonium. The yield
was 71%. Anal. Calcd (found) for [C7H7N(CH3)3]3PMo4O24: C,
28.75; H, 3.81; N, 3.28; Mo, 30.77; P, 2.53%. Found: C, 28.84;
H, 3.87; N, 3.36; Mo, 30.71; P, 2.48%. IR (neat, KBr plates):
3436, 3037, 1619, 1474, 1217, 1072, 1034, 964, 869, 780, 727,
702, 658, 588, 541 cmꢃ1
.
General Procedure for the Benzalcohol Oxidation. After 20
mmol of olefins was dissolved in 40 mL of CH3CN solvent; 15
mmol of 30% H2O2 and 0.1 mmol of catalyst were added. The
ꢂ
reaction was maintained at 70 C with vigorous stirring until the
Published on the web August 4, 2005; DOI 10.1246/bcsj.78.1575

 Mingqiang, Li
Mingqiang, Li
 Xigao, Jian
Xigao, Jian