115436-72-1 Usage
Description
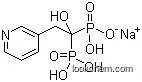
Sodium risedronate, also known as risedronic acid, is a third-generation nitrogen-containing bisphosphonate that inhibits osteoclast-mediated bone resorption and modulates bone metabolism. It is a fine white to off-white crystalline powder and is marketed under the brand name Actonel.
Used in Pharmaceutical Industry:
Sodium risedronate is used as a medication for the treatment of postmenopausal osteoporosis to reduce the risk of vertebral fractures, treatment of established postmenopausal osteoporosis to reduce the risk of hip fractures, treatment of osteoporosis in men at high risk of fractures, and treatment of Paget's disease.
Used in Antibacterial Applications:
Sodium risedronate is used as an antibacterial agent, although the specific application reason is not provided in the materials.
Sodium risedronate was launched as Actonel in the US for the treatment of Paget's disease. It is an orally active bisphosphonate that shows more potent bone antiresorptive properties than most prior bisphosphonates like etidronate. The biological mechanism by which Sodium risedronate and generally bisphosphonates act is not quite clear presently; however, recent advances have suggested that aminobisphosphonates interfere with the HMG-CoA pathway and inhibit protein prenylation, causing apoptosis of osteoclasts and macrophages. These apoptotic effects are correlated to the anti-resorptive properties of this class of compounds. In a 2-month clinical trial involving patients with Paget's disease, 30 mg Sodium risedronate daily achieved an appreciable reduction of serum alkaline phosphatase levels, an indicator of bone stabilization, in 77% of patients, compared with 11% on etidronate at 400 mg daily for 6 months. Sodium risedronate is also in development for the treatment and prevention of various bone diseases, particularly post-menopausal and corticosteroid-induced osteoporosis.
References
[1] https://www.medicines.org.uk
[2] http://www.rxlist.com
[3] http://www.healthline.com
Originator
Procter Gamble (US)
Hazard
Human systemic effects.
Biological Activity
Orally active biphosphonate that inhibits farnesyl diphosphate (FPP) synthase. Exhibits antiproliferative and proapoptotic activity in numerous tumor cell lines and inhibits osteoclast-mediated bone reabsorption in vivo .
Biochem/physiol Actions
Risedronate sodium is a bisphosphonate bone resorption inhibitor. It has an affinity for hydroxyapatite crystals in bone and acts as an antiresorptive agent and is an inhibitor of farnesyl diphosphate (FPP) synthase, which results in downstream inhibition of osteoclast activity and reduced bone resorption and turnover. Risedronate sodium has been used to treat postmenopausal osteoporosis and Paget′s disease.
Clinical Use
Bisphosphonate:
Treatment and prevention of osteoporosis (including
corticosteroid induced)
Paget’s disease
Drug interactions
Potentially hazardous interactions with other drugs
Calcium-containing substances: avoid for 2 hours
before and after administration.
Metabolism
The mean bioavailability of risedronate is 0.63% in
the fasting state, and there is no evidence of systemic
metabolism of risedronate sodium.
About half of the absorbed portion is excreted in the
urine within 24 hours; the remainder is sequestered
to bone for a prolonged period. Unabsorbed drug is
eliminated unchanged in the faeces.
Check Digit Verification of cas no
The CAS Registry Mumber 115436-72-1 includes 9 digits separated into 3 groups by hyphens. The first part of the number,starting from the left, has 6 digits, 1,1,5,4,3 and 6 respectively; the second part has 2 digits, 7 and 2 respectively.
Calculate Digit Verification of CAS Registry Number 115436-72:
(8*1)+(7*1)+(6*5)+(5*4)+(4*3)+(3*6)+(2*7)+(1*2)=111
111 % 10 = 1
So 115436-72-1 is a valid CAS Registry Number.
InChI:InChI=1/C7H11NO7P2.Na/c9-7(16(10,11)12,17(13,14)15)4-6-2-1-3-8-5-6;/h1-3,5,9H,4H2,(H2,10,11,12)(H2,13,14,15);