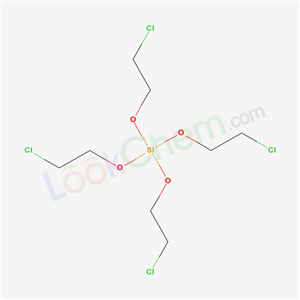
- Chemical Name:Tetrakis(2-chloroethyl) silicate
- CAS No.:18290-84-1
- Molecular Formula:C8H16Cl4O4Si
- Molecular Weight:346.11
- Hs Code.:2915900090
- DSSTox Substance ID:DTXSID70939656
- Nikkaji Number:J87.076E
- Wikidata:Q82916211
- Mol file:18290-84-1.mol
Synonyms:Tetrakis(2-chloroethyl) silicate;18290-84-1;Tetrachloroethoxysilicon;tetrakis(2-chloroethoxy)silane;BRN 1710043;Silicic acid (H4SiO4), tetrakis(2-chloroethyl) ester;AI3-08672;Ethanol, 2-chloro-, silicate;Silicic acid, tetrakis(2-chloroethyl) ester;Ethanol, 2-chloro-, tetraester with silicic acid (H4SiO4);tetrakis(2-chloroethyl) orthosilicate;C8H16Cl4O4Si;Tetra(2-chloroethoxy)silane;C8-H16-Cl4-O4-Si;SCHEMBL4452327;DTXSID70939656;Ethanol, 2-chloro-, silicate (6CI);LS-145323;Ethanol, 2-chloro-, tetraester with silicic acid (H4SiO4) (8CI)