GModel
CATTOD-8841; No. of Pages11
ARTICLE IN PRESS
2
D. Gudarzi et al. / Catalysis Today xxx (2014) xxx–xxx
employed as support. Palladium (II) chloride was used as metal
precursors.
The activated carbon cloth was first treated in an oven at 100 ◦C
for about 12 h. Then chemical cleaning was accomplished by a 50%
solution of methanol at room temperature for 1 h, and then washed
with plenty of deionized water. In order to oxidize activated carbon
cloth, the cleaned ACC was treated with a 20% solution of nitric
or acetic acid at room temperature for 40 h. After this oxidation
treatment the samples were washed with deionized water until
These samples are referred as oxidized ACC. Acid pre-treatment
step was used to create oxygen-containing functional groups on the
surface of activated carbon and to modify the surface as suggested
by several authors [40–45].
Pd catalysts on non-oxidized and oxidized activated carbon
cloth were prepared by impregnation of the supports with an aque-
ous acidic solution of PdCl2 at room temperature for 6–7 h. The
impregnation was done in a glass reactor with stirring rate of
600–700 rpm. The ratio of the solution volume to catalyst mass was
500 ml/g. The concentration of the metal precursor between 16.67
and 83.3 mg/L for 1 g of the support gave palladium loading in the
range 1–5 wt%.
After impregnation, the catalysts were left at room tempera-
ture for 3 h, and then dried at 60 ◦C over night. To investigate the
effects of the oxidation state of the metallic phase on the decompo-
sition/hydrogenation activity, the dried fresh catalysts were finally
treated with different processes. The reduced samples were pre-
pared by treating the fresh dried catalysts with H2 at 185 ◦C under
3.5 bar for around 12 h. A series of different calcined samples were
prepared by oxidizing the fresh dried catalysts at temperatures 135,
185, and 235 ◦C in static air for 12 h. The calcination temperatures
were selected on the basis of our earlier study [8] in order to be in
the safe thermal treatment region of the supports and their surface
functional groups.
Fig. 1. Reaction pathways involved in the direct reaction of H2 and O2.
active and selective than basic catalysts (Pd/Al2O3) [10–12]. H2O2
acidic medium [13]. The H2O2 decomposition activity of the sup-
ported Pd catalysts is strongly influenced by the oxidation state
of Pd in the catalysts. The supported palladium oxide (PdO) cata-
lysts have shown much lower H2O2 decomposition activity than
destruction (Fig. 1, reactions iii and iv) activity of Pd-based catalysts
decreases drastically due to the addition of different halide anions
(Cl−, Br− or I−) in the catalyst [4] or in the acidic reaction medium
[14–28]. However, the presence of halide ions together with H+ ions
in a reaction medium may cause the reaction medium to become
strongly corrosive toward metallic reactor materials, particularly
stainless steel. In addition, the halide ions together with H+ ions
form a complex reaction mixture from which the isolation of H2O2
and catalyst recovery is a difficult task [1]. Fu et al. [29] reported that
position of H2O2.
ladium and platinum in liquid phase, due to their various industrial
such as activated carbon cloths and activated carbon felts, repre-
sent noticeable advantageous characteristics with respect to the
usual granular activated carbons [33]. High surface area is normally
in the range of 1500–3000 m2 g−1 [34], and the porous network is
mainly formed by deep pores in a narrow range of sizes, especially
micropores [35]. Because of mechanical elasticity and geometric
flexibility, fibrous cloths can be easily bent and rolled to fit in par-
ticular uses. Cloths woven from thin m-sized fibers reduce the
and in multi-phase reactors where one or more dissolved species
have to react with gaseous compounds of limited solubility [36].
Fast adsorption/desorption rates have been claimed as an addi-
tional advantage [37,38]. Moreover, in our microstructured reactor
[39], ACC catalyst is an attractive alternative because it can be
replaced quickly and easily when needed. So far, the use of text-
ural form of activated carbons (ACC and ACF) as catalyst supports
is not well covered in scientific literature.
Pore size and surface area of different samples were measured
(Sorptomatic 1900, Carlo Erba Instruments). All samples were
degassed at a temperature of 100 ◦C for about 3 h prior to the mea-
surements. The results have been presented in our previous work
Atomic absorption spectroscopy (AAS) was used to determine
the wt% of the metal incorporated into the support after impregna-
tion. AAS was performed with a Thermo Scientific ICE 3000 series
atomic absorption spectrometer using an air–acetylene flame. Sam-
ples for analysis were prepared by dissolving 200 mg of the dried
catalyst in an aqua regia solution, followed by the addition of 500 ml
deionized water to dilute the sample.
Temperature programmed desorption (TPD) was employed to
investigate surface chemistry of carbon fibers in samples. The TPD
tests were conducted in a set-up made of a U-shaped tubular reac-
tor, placed inside an electrical furnace coupled to a Blazers Omnistar
mass spectrometer for gas analysis. 200 mg of samples were heated
up to 1000 ◦C with a constant heating rate of 50 ◦C/min. During the
tests, He was passed through the reactor with the flow rate equal
to 60 ml/min. The amounts of CO and CO2 desorbed from samples
were recorded by the mass spectrometer.
The preparation of Pd catalysts on activated carbon cloth for the
direct synthesis was thoroughly studied in our previous work [8].
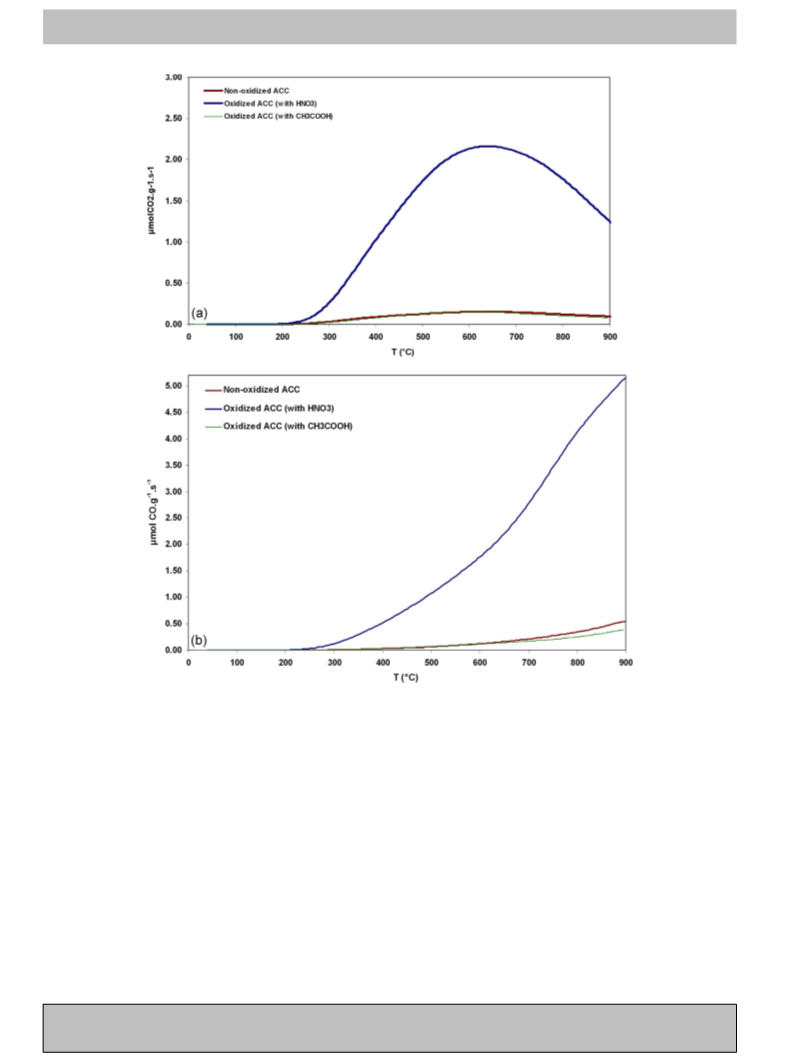
Since H2O2 decomposition can result in the loss of selectivity and
yield, it is important to have a deep understanding of the factors
affecting the H2O2 decomposition reactions (Fig. 1, reactions iii and
iv). Destruction of H2O2 is a complex process and could be affected
by different factors. In the present work, the effects of the reaction
The goal of this paper and our pervious work [8] is to develop
a selective Pd catalyst supported on activated carbon cloth for the
direct synthesis. This catalyst will be used in a novel microstruc-
tured reactor [39].
2. Experimentation
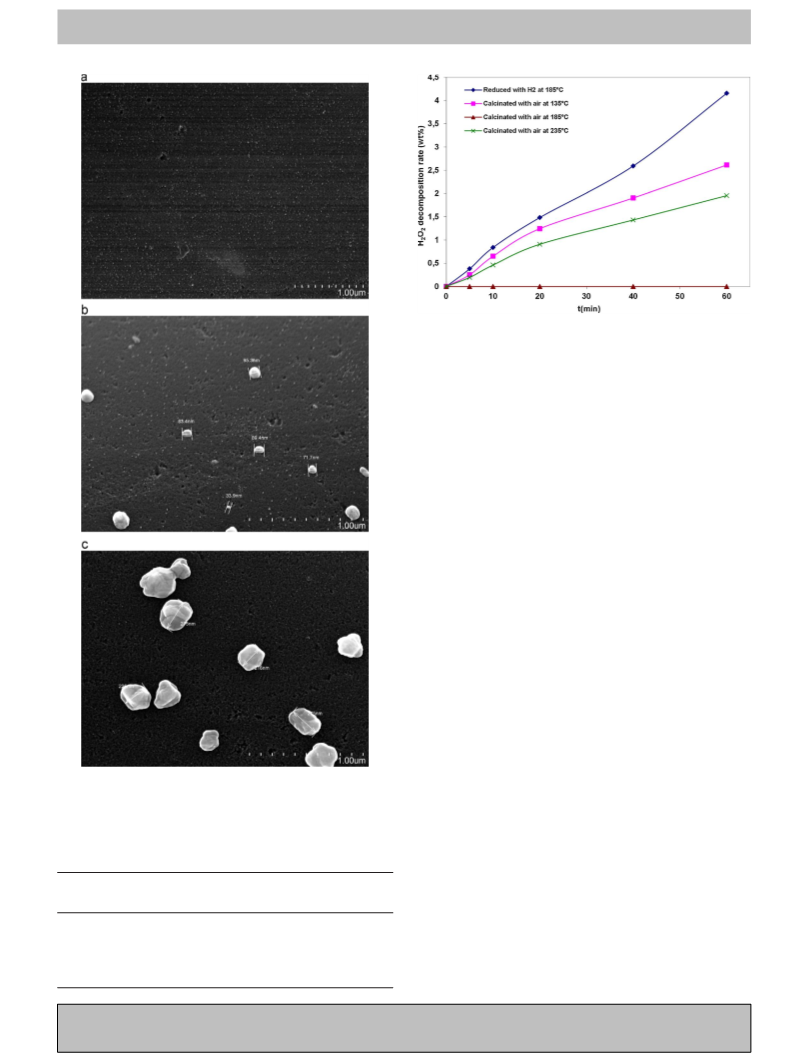
Ultra-High Resolution Field Emission Scanning Electron Micro-
scope (UHRFESEM) was used to evaluate particle size, shape,
surface morphology, and to monitor the agglomeration tendency
of metal particles. The UHRFESEM and Energy Dispersive Spec-
troscopy (EDS) analysis were done by a Hitachi S-4800 microscope
with an X-ray detector for micro-analytical X-ray mapping and
2.1. Catalysts preparation
A commercial activated carbon cloth ACC-5092-20 (Kynol
Europa GmbH), after a cleaning and a chemical treatment, was
Please cite this article in press as: D. Gudarzi, et al., Factors affecting catalytic destruction of H2O2 by hydrogenation and decomposition over Pd

 Gudarzi, Davood
Gudarzi, Davood
 Ratchananusorn, Warin
Ratchananusorn, Warin
 Turunen, Ilkka
Turunen, Ilkka
 Heinonen, Markku
Heinonen, Markku
 Salmi, Tapio
Salmi, Tapio