
Bulletin of the Chemical Society of Japan p. 3295 - 3300 (1988)
Update date:2022-08-11
Topics:
 Matsui, Hiroshi
Matsui, Hiroshi
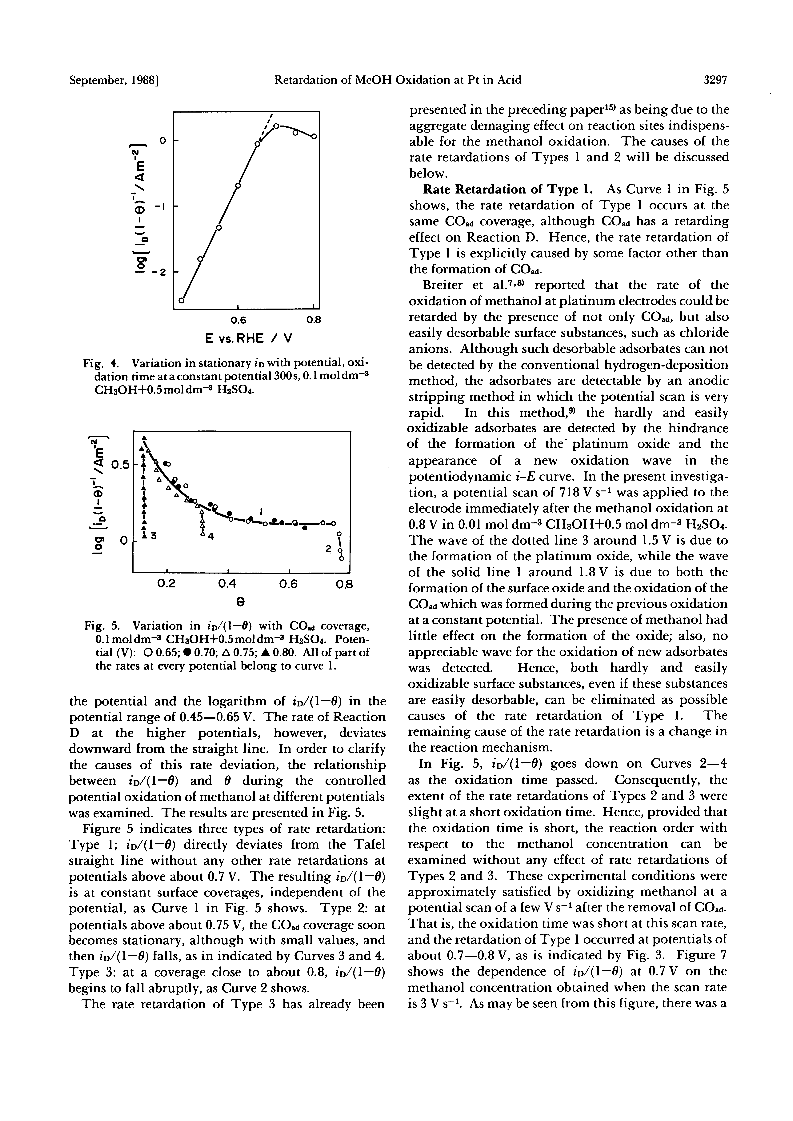
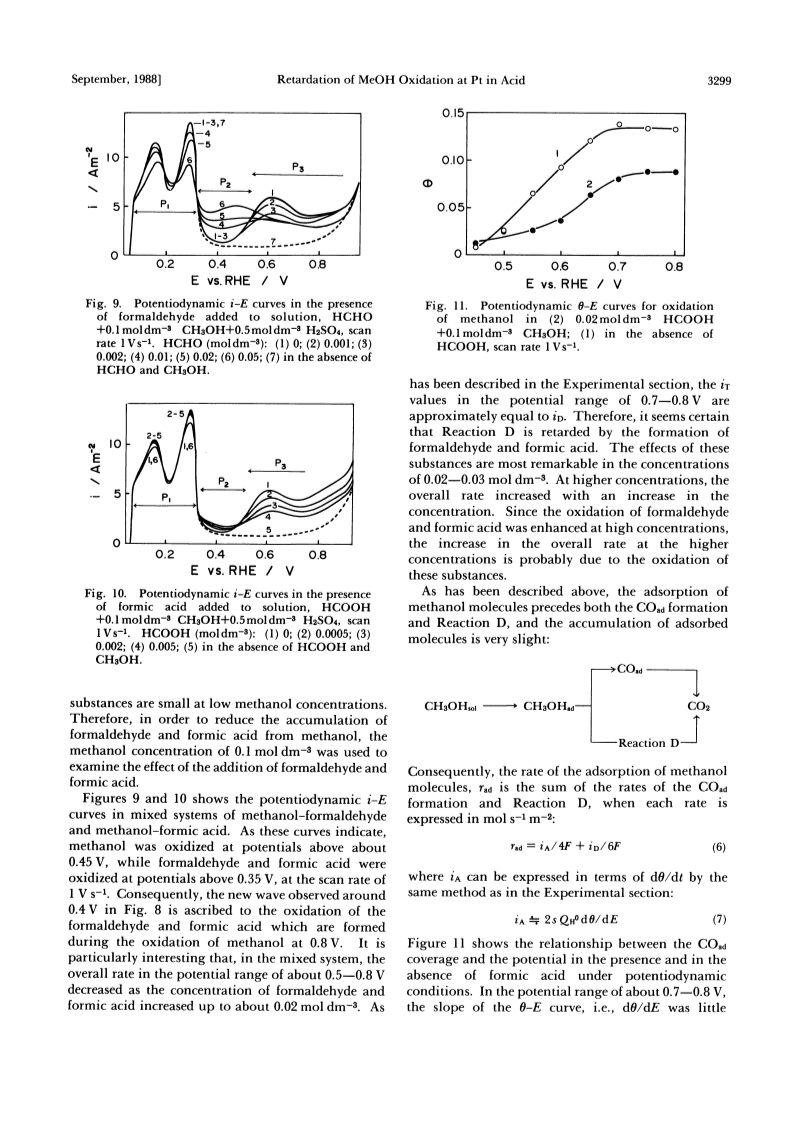
The rate retardation of the oxidation of methanol at the potential range of about 0.65-0.8 V vs. a reversible hydrogen electrode on a platinum electrode in 0.5 mol dm-3 H2SO4 was studied.The rate retardation of the overall oxidation was caused by that of the oxidation, Reaction D, not via COad.From the relationship among the rate of Reaction D, the COad coverage, and the potentials, three types of rate retardation were found out: Type 1-Reaction D is not accelerated by the potential, and the rate of the reaction is determined by the COad coverage and the methanol concentration.Type 2- the rate of Reaction D decreases at stationary COad coverages as the oxidation is prolonged.Type 3- the rate decreases at COad coverages close to the limiting value.It is proposed that Types 1 and 2 of the rate retardations take place when the adsorption of methanol molecules is rate-determining, and when the formaldehyde and formic acid formed from methanol are accumulated in the vicinity of the electrode, respectively.Type 3 of the rate retardation has been explained in a preceding paper in terms of the aggregate damaging effect of COad.
View More



Hangzhou Pharma & Chem Co.,Ltd.
Contact:+86-571-87040515
Address:No,139Qingchun Rd
Nanjing Habo Medical Technology Co., Ltd.
Contact:025-85769882
Address:No.108. Ganjiabian east. Qixia District .Nanjing
Contact:86+21-56421993
Address:3F,BUILDING 10,NO.2889 JINKE ROAD, SHANGHAI.
Zhejiang Kente Chemical Co.,Ltd.
Contact:86-0576-87651912
Address:No.7, Fengxi West Road, Modern Industrial Zone
HEZE KINGVOLT CHEMICAL CO., LTD
Contact:86-573-82118911
Address:Juancheng Industry Park
Doi:10.1016/0040-4039(95)00711-K
(1995)Doi:10.1038/s41557-019-0304-z
(2019)Doi:10.1039/P29760001342
(1976)Doi:10.3762/bjoc.13.98
(2017)Doi:10.1016/S0040-4039(00)70652-6
(1989)Doi:10.1021/jo00898a038
(1975)