Organic Process Research & Development 2010, 14, 252–258
Full Papers
Direct Oxidation of Cyclohexene with Inert Polymeric Membrane Reactor
Maria G. Buonomenna,*,† Giovanni Golemme,†,‡ Maria P. De Santo,§ and Enrico Drioli†,‡
Department of Chemical, Engineering and Materials, UniVersity of Calabria and Consortium INSTM, ITM-CNR, and
CEMIF.CAL, CNR-Licryl Laboratory, Physics Department, UniVersity of Calabria, Via Pietro Bucci,
87030 Rende (Cosenza), Italy
Scheme 1. Pathway for the synthesis of adipic acid by
Abstract:
oxidation of cyclohexene with hydrogen peroxide6
In this work, the use of PVDF flat membranes as contactors for
direct solvent-free biphasic oxidation of cyclohexene to adipic acid
has been reported. The oxidation has been carried out using 30%
H2O2 and ammonium molybdate ((NH4)6Mo7O24) in the
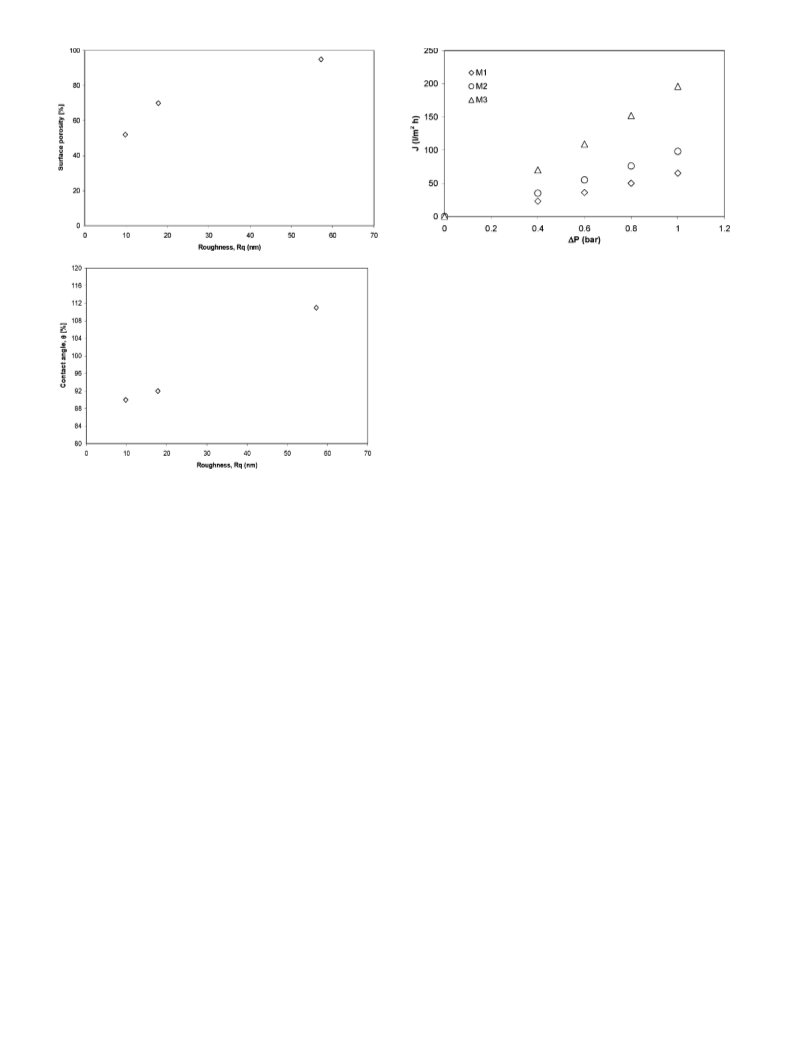
presence of succinic acid. The effect of different membranes
as interfaces between the organic phase, containing cyclo-
hexene, and the aqueous phase, with the oxidant and
catalyst, has been studied and related to conversion and
selectivity.
it is possible to achieve simultaneous, end-on oxidation of both
terminal methyl ends of n-hexane, using air as an oxidant under
moderate conditions.
Research on the clean synthesis of adipic acid has been
reported by Sato et al.:6 cyclohexene has been efficiently
oxidized to adipic acid with aqueous 30% H2O2 in the presence
of small amounts of Na2WO4 ·2H2O and [CH3(n-C8H17)3N]-
HSO4 as a phase-transfer catalyst.
1. Introduction
Adipic acid (AA) is of great importance commercially in
that it is used in the manufacture of nylon 6,6, which is
extensively used in many products such as tire reinforcements,
adhesives, upholstery, specialty foams, carpet fibers and several
items of clothing.1 Currently, its main method of manufacture
is a costly, multistep process involving homogeneous catalysts
and aggressive oxidants such as concentrated nitric acid. The
scientific challenge addressed by this work is to devise a one-
step synthesis operating in an environmentally friendly, solvent-
free, biphasic or monophasic system using green co-oxidants
such as oxygen (air) or hydrogen peroxide.2,3
Dugal et al.4 have shown that, with microporous FeAlPO-
31 as a heterogeneous catalyst, a one-step, solvent-free conver-
sion of cyclohexene to AA may be effected at 100-130 °C
and 1.5 MPa of air.
Raja et al.5 have shown that it is also possible to produce
AA starting from a linear hydrocarbon such as n-hexane. By
using a heterogeneous aluminophosphate catalyst (AlPO-18),
typically having a cobalt:phosphorous ratio of 0.08 or greater,
Cyclohexene (CH) is transformed into adipic acid (AA)
in a “one-pot” synthesis following the reaction shown in
Scheme 1.
In the pathway proposed by Sato et al.6 the controlling step
is the hydrolysis of cyclohexene oxide to 1,2-cyclohexandiol
under acidic conditions.
Deng et al.7 reported a cheaper and environmentally benign
peroxytungstate-organic complex which could be used as a
highly efficient catalyst for the direct catalytic oxidation of
cyclohexenes to adipic acid with hydrogen peroxide without
any phase transfer catalyst.
The peroxytungstate was complexed with organic acids, and
the resultant catalyst system not only possessed the capability
of carrying active oxygen species but also became oleophilic;
therefore, a very good catalyst system for the oxidation of
cyclohexene could be formed. Similar to peroxytungstate, when
the peroxymolybdate was complexed with the organic acid, it
became oleophilic, and the substrate/catalyst ratio could be
raised to about 1000, whereas it is 40 for analogous biphasic
systems.7,8
In recent years, surfactant-type catalysts (STC) containing
polyoxoperoxo metalates have been successfully used on various
typical organic synthetic reactions with hydrogen peroxide.9 In
principle, through combining the anion of polyoxometalate with
surfactant to form a STC and tuning the hydrophile-lipophile
balance of surfactant, the amphiphilic catalyst can be adapted
* Corresponding author. E-mail: mg.buonomenna@unical.it.
† Department of Chemical, Engineering and Materials, University of Calabria
and Consortium INSTM.
‡ ITM-CNR.
§ CEMIF.CAL, CNR-Licryl Laboratory, Physics Department, University of
Calabria.
(1) Davis, D. D.; Kemp, D. R. Kirk-Othmer Encyclopedia of Chemical
Technology; Wiley-VCH Verlag: New York, 1991.
(2) D’Alessandro, N.; Liberatore, L.; Tonucci, L.; Morvillo, A.; Bressan,
M. New J. Chem. 2001, 25, 1319–1324.
(3) Centi, G.; Perathoner, S. Catal. Today 2003, 77, 287–297.
(4) Dugal, M.; Sankar, G.; Raja, R.; Thomas, J. M. Angew. Chem., Int.
Ed. 2000, 39, 2310.
(6) Sato, K.; Aoki, M.; Noyori, R. Science 1998, 281, 1646–1647.
(7) Deng, Y.; Ma, Z.; Wang, K.; Chen, J. Green Chem. 1999, 1, 275–
276.
(5) Raja, R.; Sankar, G.; Thomas, J. M. Angew. Chem., Int. Ed. 2000, 39,
2313–2313.
(8) Jiang, H.; Gong, H.; Yang, Z.; Zhang, X.; Sun, Z. React. Kinet. Catal.
Lett. 2002, 75, 315–321.
252
•
Vol. 14, No. 1, 2010 / Organic Process Research & Development
10.1021/op900022t CCC: $40.75 2010 American Chemical Society
Published on Web 11/23/2009

 Buonomenna, Maria G.
Buonomenna, Maria G.
 Golemme, Giovanni
Golemme, Giovanni
 De Santo, Maria P.
De Santo, Maria P.
 Drioli, Enrico
Drioli, Enrico