Chemical Science
ARTICLE
DOI: 10.1039/C6SC02842J
Journal Name
one can expect that ER based process could lead to similar high 11. N. Z. Xie, H. Liang, R. B. Huang and P. Xu, Biotechnol. Adv., 2014, 32,
6
15ꢀ622.
2. J.ꢀL. Yu, X.ꢀX. Xia, J.ꢀJ. Zhong and Z.ꢀG. Qian, Biotechnol. Bioeng.,
014, 111, 2580ꢀ2586.
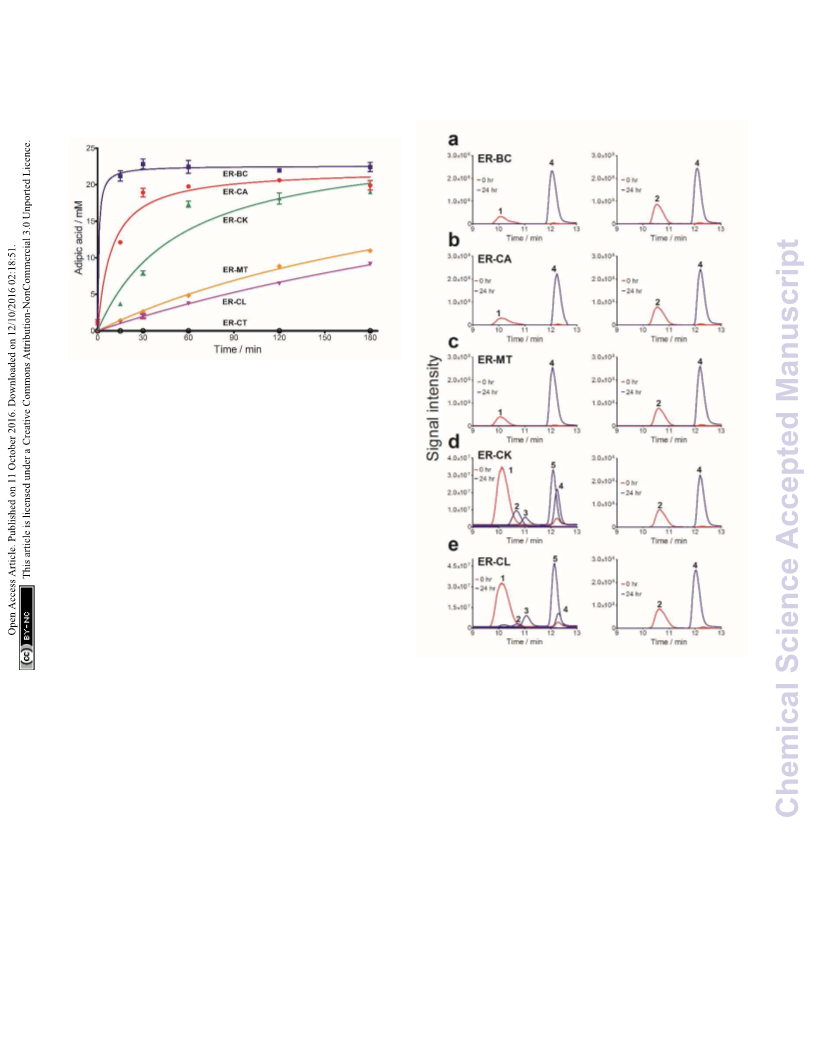
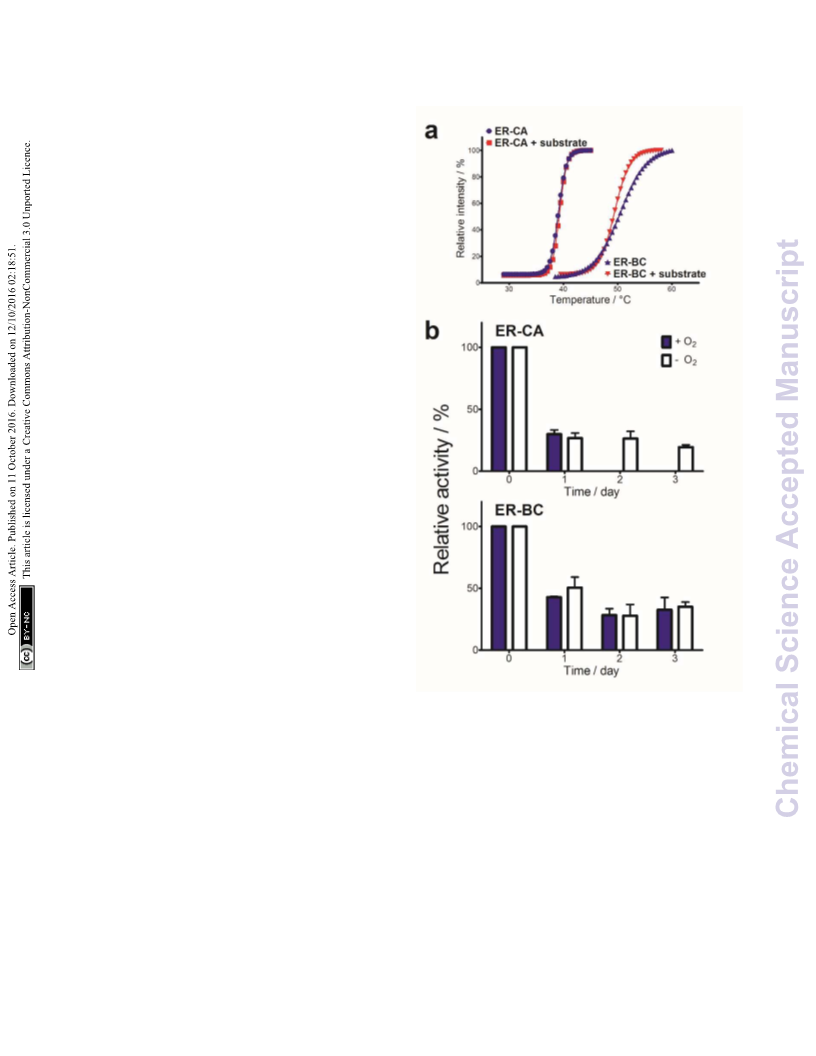
titers. In particular, ERꢀBC exhibits high oxygen tolerance and
thermostability making it useful for in vivo and in vitro
applications to overcome limitations of chemical catalysts.
Thus, the C=C hydrogenation activity of ERs demonstrated in
our work can potentially replace the chemical hydrogenation
step in current synthetic protocols creating a completely bioꢀ
1
1
2
3. D. G. M., M. V. and B. T. R., in Modern Applications of High
Throughput R&D in Heterogeneous Catalysis, eds. H. A. and V. A. F.,
Bentham Science, 2014, DOI: 10.2174/97816080587231140101, ch. 8,
pp. 288ꢀ309.
based pathway for greener production of adipic acid or other 14. J. M. Clomburg, M. D. Blankschien, J. E. Vick, A. Chou, S. Kim and R.
biochemicals e.g. 3ꢀphenylpropanoic acid from renewable
feedstocks.
Gonzalez, Metab. Eng., 2015, 28, 202ꢀ212.
5. S. Cheong, J. M. Clomburg and R. Gonzalez, Nat. Biotech., 2016, 34,
1
1
5
56ꢀ561.
6. K. A. Curran, J. M. Leavitt, A. S. Karim and H. S. Alper, Metab. Eng.,
013, 15, 55ꢀ66.
Acknowledgements
2
The authors thank all members of the BioZone Centre for Applied 17. H.ꢀM. Jung, M.ꢀY. Jung and M.ꢀK. Oh, Appl. Microbiol. Biotechnol.,
Science and Bioengineering and Christine Achampong for help in
2015, 99, 5217ꢀ5225.
conducting the experiments. Dr. K. T. Shanmugam (University of 18. D. R. Vardon, N. A. Rorrer, D. Salvachua, A. E. Settle, C. W. Johnson,
M. J. Menart, N. S. Cleveland, P. N. Ciesielski, K. X. Steirer, J. R.
Dorgan and G. T. Beckham, Green Chem., 2016, 18, 3397ꢀ3413.
9. D. R. Vardon, M. A. Franden, C. W. Johnson, E. M. Karp, M. T.
Guarnieri, J. G. Linger, M. J. Salm, T. J. Strathmann and G. T. Beckham,
Energ. Envrion. Sci., 2015, 8, 617ꢀ628.
Florida) and Dr. P. Hallenbeck (University of Montreal) are thanked
for providing the B. coagulans 36D1 genomic DNA and the E. coli
BL21 (DE3) ∆iscR strain, respectively. This work was supported by
the Government of Canada through Genome Canada and the Ontario
Genomics Institute (2009ꢀOGIꢀABCꢀ1405), Ontario Research Fund
1
2
0. United States of America Pat., US8592189 B2, 2013.
(
ORFꢀGL2ꢀ01ꢀ004), NSERC Strategic Project Grant, and 21. J. C. J. Bart and S. Cavallaro, Ind. Eng. Chem. Res., 2015, 54, 567ꢀ576.
BiofuelsNet, by Industrial Strategic Technology Development 22. G. Sirasani, L. Tong and E. P. Balskus, Angew. Chem. Int. Ed., 2014, 53,
Program (10047910, Production of biobased cadaverine and
polymerization of Bioꢀpolyamide 510) funded by the Ministry of
Trade, Industry & Energy (MOTEI, Korea).
7785ꢀ7788.
2
2
3. A. J. J. Straathof, Chem. Rev., 2014, 114, 1871ꢀ1908.
4. Y. Deng, L. Ma and Y. Mao, Biochem. Eng. J., 2016, 105, Part A, 16ꢀ
2
6.
2
5. X. Li, D. Wu, T. Lu, G. Yi, H. Su and Y. Zhang, Angew. Chem. Int. Ed.,
2014, 53, 4200ꢀ4204.
Notes and references
Center for Bioꢀbased Chemistry, Division of Convergence Chemistry,
a
26. H. Simon, J. Bader, H. Guenther, S. Neumann and J. Thanos, Angew.
Chem. Int. Ed., 1985, 24, 539ꢀ553.
Korea Research Institute of Chemical Technology, 141 Gajeongꢀro,
Yuseongꢀgu, Daejeon 34114, Republic of Korea.
2
2
2
7. H. S. Toogood, J. M. Gardiner and N. S. Scrutton, ChemCatChem, 2010,
2, 892ꢀ914.
8. C. K. Winkler, G. Tasnádi, D. Clay, M. Hall and K. Faber, J.
Biotechnol., 2012, 162, 381ꢀ389.
b
Department of Chemical Engineering and Applied Chemistry,
University of Toronto, 200 College Street, ON M5S 3E5, Canada.
9. R. Stuermer, B. Hauer, M. Hall and K. Faber, Curr. Opin. Chem. Biol.,
c
Institute of Biochemistry, Department of Biotechnology & Enzyme
2
007, 11, 203ꢀ213.
Catalysis, Greifswald University, FelixꢀHausdorffꢀStrasse 4, 17487 30. N. Iqbal, F. Rudroff, A. Brigé, J. Van Beeumen and M. D. Mihovilovic,
Tetrahedron, 2012, 68, 7619ꢀ7623.
1. A. Z. Walton, B. Sullivan, A. C. PattersonꢀOrazem and J. D. Stewart,
ACS Catal., 2014, 4, 2307ꢀ2318.
Greifswald, Germany.
3
‡
*
These authors contributed equally to this work.
Corresponding authors: Jeong Chan Joo, Eꢀmail: jcjoo@krict.re.kr; 32. G. Steinkellner, C. C. Gruber, T. PavkovꢀKeller, A. Binter, K. Steiner, C.
Winkler, A. Łyskowski, O. Schwamberger, M. Oberer, H. Schwab, K.
Faber, P. MacHeroux and K. Gruber, Nat. Commun., 2014, 5.
3. A. B. Daugherty, S. Govindarajan and S. Lutz, J. Am. Chem. Soc., 2013,
3
3
3
1
35, 14425ꢀ14432.
4. T. Reß, W. Hummel, S. P. Hanlon, H. Iding and H. Gröger,
ChemCatChem, 2015, 7, 1302ꢀ1311.
5. M. Gall, M. Thomsen, C. Peters, I. V. Pavlidis, P. Jonczyk, P. P. Grunert,
S. Beutel, T. Scheper, E. Gross, M. Backes, T. Geißler, J. P. Ley, J. M.
Hilmer, G. Krammer, G. J. Palm, W. Hinrichs and U. T. Bornscheuer,
Angew. Chem. Int. Ed., 2014, 53, 1439ꢀ1442.
†
Electronic Supplementary Information (ESI) available: [Experimental
details and supplementary figures and tables]. See
DOI: 10.1039/b000000x/
1
2
.
.
M. Okada, Prog. Polym. Sci., 2002, 27, 87ꢀ133.
M. Kalim Akhtara, N. J. Turner and P. R. Jones, Proc. Natl. Acad. Sci. 36. H. S. Toogood and N. S. Scrutton, Catal. Sci. Technolog., 2013, 3, 2182ꢀ
U. S. A., 2013, 110, 87ꢀ92.
2194.
3
7. F. Rohdich, A. Wiese, R. Feicht, H. Simon and A. Bacher, J. Biol.
Chem., 2001, 276, 5779ꢀ5787.
3
4
5
6
.
.
.
.
J. Nielsen, M. Fussenegger, J. Keasling, S. Y. Lee, J. C. Liao, K. Prather
and B. Palsson, Nat. Chem. Biol., 2014, 10, 319ꢀ322.
H. C. Tseng and K. L. J. Prather, Proc. Natl. Acad. Sci. U. S. A., 2012, 38. P. A. Hubbard, X. Liang, H. Schulz and J. J. P. Kim, J. Biol. Chem.,
2
003, 278, 37553ꢀ37560.
1
09, 17925ꢀ17930.
J. W. Song, E. Y. Jeon, D. H. Song, H. Y. Jang, U. T. Bornscheuer, D. K. 39. H. Simon, J. Bader, H. Guenther, S. Neumann and J. Thanos, Angew
Oh and J. B. Park, Angew. Chem. Int. Ed., 2013, 52, 2534ꢀ2537.
N. Oberleitner, C. Peters, J. Muschiol, M. Kadow, S. Saß, T. Bayer, P.
Schaaf, N. Iqbal, F. Rudroff, M. D. Mihovilovic and U. T. Bornscheuer,
ChemCatChem, 2013, 5, 3524ꢀ3528.
Chem (Int Ed Engl), 1985, 24, 539ꢀ553.
4
0. C. Stueckler, M. Hall, H. Ehammer, E. Pointner, W. Kroutil, P.
Macheroux and K. Faber, Org. Lett., 2007, 9, 5409ꢀ5411.
41. R. D. Finn, A. Bateman, J. Clements, P. Coggill, R. Y. Eberhardt, S. R.
Eddy, A. Heger, K. Hetherington, L. Holm, J. Mistry, E. L. L.
Sonnhammer, J. Tate and M. Punta, Nucleic Acids Res., 2014, 42, D222ꢀ
D230.
7
8
9
1
.
.
.
E. Ricca, B. Brucher and J. H. Schrittwieser, Adv. Synth. Catal., 2011,
3
53, 2239ꢀ2262.
I. OrozꢀGuinea and E. GarcíaꢀJunceda, Curr. Opin. Chem. Biol., 2013,
7, 236ꢀ249.
Y. S. Jang, B. Kim, J. H. Shin, Y. J. Choi, S. Choi, C. W. Song, J. Lee, 43. M. S. Rhee, B. E. Moritz, G. Xie, T. Glavina Del Rio, E. Dalin, H. Tice,
4
2. A. D. N. Vaz, Biochemistry, 1995, 34, 4246ꢀ4256.
1
H. G. Park and S. Y. Lee, Biotechnol. Bioeng., 2012, 109, 2437ꢀ2459.
D. Bruce, L. Goodwin, O. Chertkov, T. Brettin, C. Han, C. Detter, S.
Pitluck, M. Land, M. Patel, M. Ou, R. Harbrucker, L. O. Ingram and K.
T. Shanmugam, Stand. Genomic Sci., 2011, 5, 331–340.
0. T. Polen, M. Spelberg and M. Bott, J. Biotechnol., 2013, 167, 75ꢀ84.
6
| J. Name., 2012, 00, 1-3
This journal is © The Royal Society of Chemistry 2012

 Joo, Jeong Chan
Joo, Jeong Chan
 Khusnutdinova, Anna N.
Khusnutdinova, Anna N.
 Flick, Robert
Flick, Robert
 Kim, Taeho
Kim, Taeho
 Bornscheuer, Uwe T.
Bornscheuer, Uwe T.
 Yakunin, Alexander F.
Yakunin, Alexander F.
 Mahadevan, Radhakrishnan
Mahadevan, Radhakrishnan