
Applied Catalysis A: General p. 118 - 124 (2011)
Update date:2022-08-17
Topics:
 Cavani, Fabrizio
Cavani, Fabrizio
 Ferroni, Laura
Ferroni, Laura
 Frattini, Alessandra
Frattini, Alessandra
 Lucarelli, Carlo
Lucarelli, Carlo
 Mazzini, Andrea
Mazzini, Andrea
 Raabova, Katerina
Raabova, Katerina
 Alini, Stefano
Alini, Stefano
 Accorinti, Pasquale
Accorinti, Pasquale
 Babini, Pierpaolo
Babini, Pierpaolo
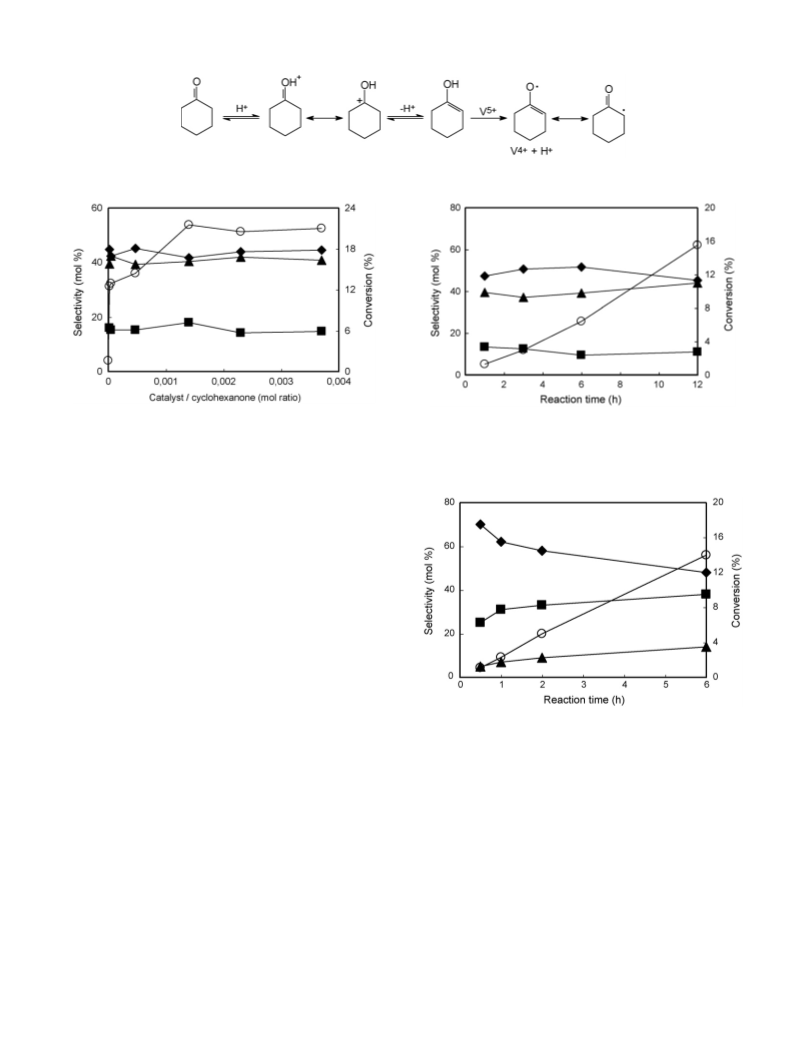
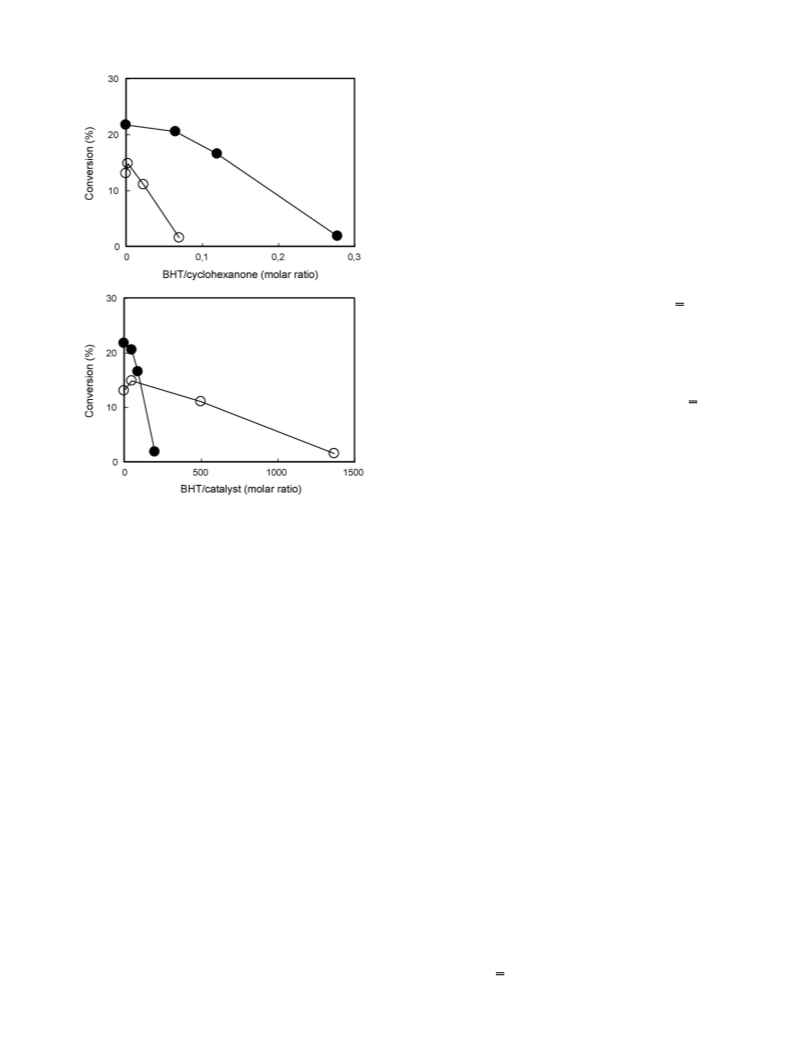
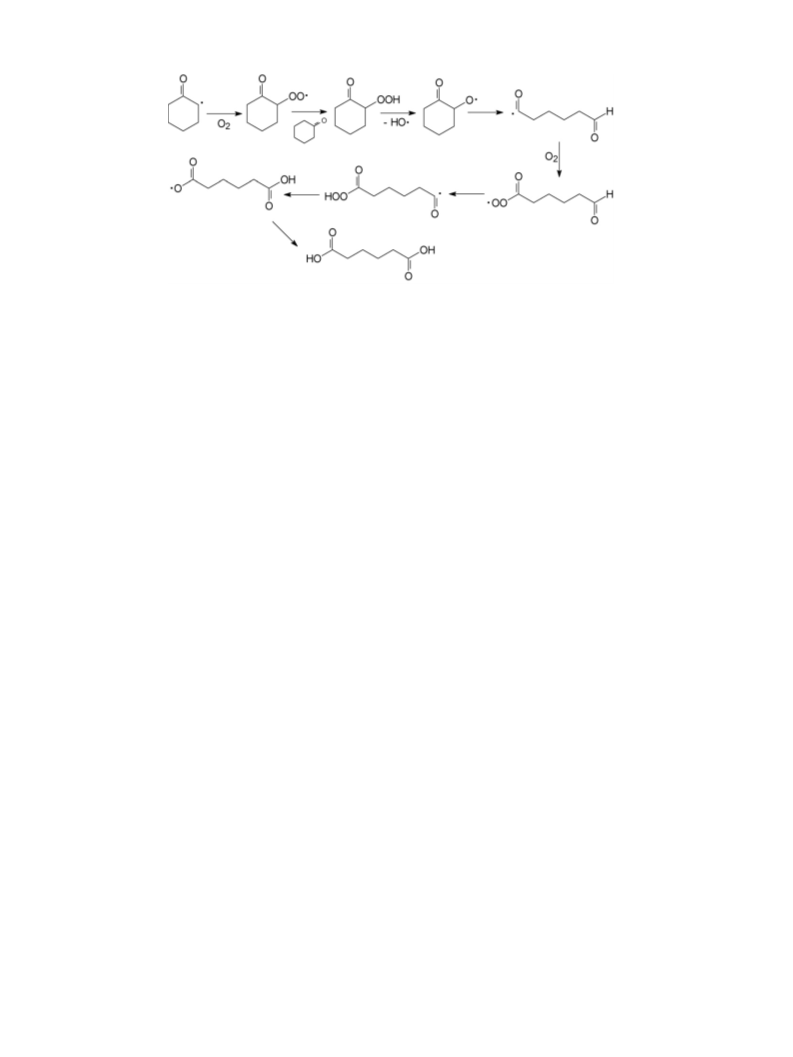
This report deals with the results of a study on the oxidation of cyclohexanone to adipic acid with air, catalysed by Keggin-type polyoxometalates of composition H3+xPMo12-xVxO40 (x = 1 and 2), which was carried out in a semi-continuous stirred-tank reactor. It was found that when conducted in the presence of a water-only solvent, the reaction proceeds with a redox mechanism, in which the step of reoxidation of the reduced POM by oxygen is rate limiting. When, however, the reaction was carried out with an acetic acid co-solvent, a radical-chain autoxidation mechanism prevailed, especially when very low amounts of catalyst were used. Autoxidation overlapped with the redox mechanism when the catalyst-to- cyclohexanone ratio was increased. Moreover, the composition of the polyoxometalate, that is, the number of V atoms per Keggin unit, affected the relative importance of the two mechanisms. The selectivity to adipic acid achieved was a function of the reaction mechanism, but also was affected by cyclohexanone conversion, due to the presence of a complex reaction network.
View More



Jiacheng-Chem Enterprises Limited
Contact:86-571-86711508
Address:19 Floor, CIBC Holley International Building, No. 198,Wuxing Road, Hangzhou, China,310020
HEZE KINGVOLT CHEMICAL CO., LTD
Contact:86-573-82118911
Address:Juancheng Industry Park
Chongqing Changfeng Chemical Co., Ltd.
website:http://www.changfengchem.com
Contact:+86-23-67896333
Address:30th Floor, Longhu Ziduxingzuo Building A, 1st Branch,YuSong Rd., Yubei District, Chongqing, China
Contact:+36(21)2523420
Address:Head office: 1102 Budapest, SZENT LASZLO TER 24/B. 1/1., HUNGARY / CHINA
Zhuzhou Farshine Chemical Industry Co., Ltd
Contact:0086-731-28482786
Address:No. 1,Shui Xian Road, He Tang Dictrict,Hunan-412000,China
Doi:10.1021/ol070240k
(2007)Doi:10.1021/om300868k
(2013)Doi:10.1016/j.ejmech.2019.04.071
(2019)Doi:10.1021/ol102208s
(2010)Doi:10.1039/b406179a
(2004)Doi:10.1134/S1070363210070327
(2010)