8
620
Z.-H. Lu et al. / Tetrahedron Letters 43 (2002) 8617–8620
References
1
. For recently reported reducing systems, see: (a) Shibata,
I.; Kawakami, T. M.; Tanizawa, D.; Suwa, T.; (b)
Sugiyama, E.; Matsuda, H.; Baba, A. J. Org. Chem.
1
998, 63, 383; Shimizu, M.; Sahara, T.; Hayakawa, R.
Chem. Lett. 2001, 792; (c) Knettle, B. W.; Flowers, R.
A., II Org. Lett. 2001, 3, 2321.
. Reductions in Organic Synthesis: Recent Advances and
Practical Applications; Abdel-Magid, Ahmed F., Ed.;
Chapter 11 and references cited therein.
1
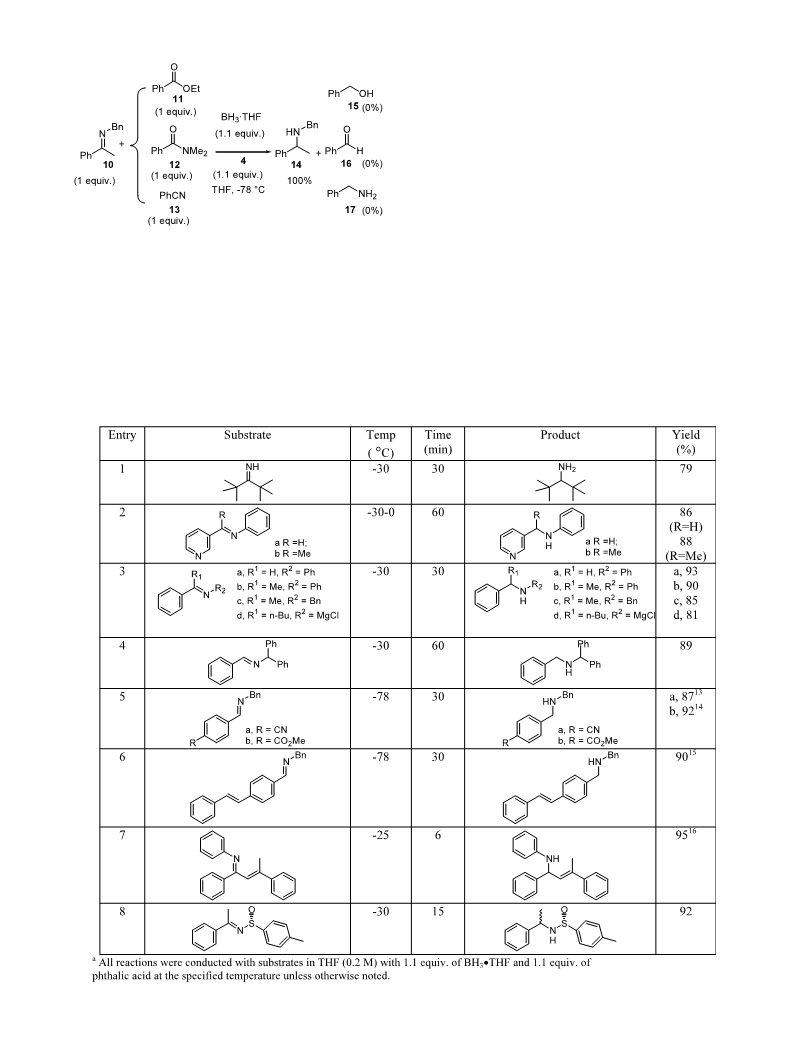
3. General procedure for the reduction of imines by borane
and phthalic acid. The preparation of 4-(benzylamino-
methyl)-benzonitrile (entry 6, Table 3) was described as
an example. Phthalic acid (365.5 mg, 2.2 mmol) was
added to a solution of imine (440.5 mg, 2.0 mmol) in 8
mL THF at 25°C. After cooling the solution to −78°C,
2
3
. For a review of the chemistry of sodium cyanoborohy-
dride, see: Synthesis 1975, 135.
4
. Reductions in Organic Synthesis: Recent advances and
practical applications; Abdel-Magid, Ahmed F., Ed.;
Chapter 12 and references cited therein.
. The Sigma-Aldrich Library of Chemical Safety Data, 1st
ed.; Lenga, R. E., Ed.; Sigma-Aldrich Corp., 1985; p.
BH ·THF (2.2 mL, 1 M in THF, 2.2 mmol) was added
3
below −70°C or at the specified temperature. Reaction
was stirred for 30 min and quenched with methanol (2
mL). It was then allowed to warm to room temperature.
Isopropyl acetate (25 mL) was added and the reaction
was washed with 1 M KOH (3×20 mL), water (2×25 mL)
and dried. Solvents were evaporated in vacuo to give the
secondary amines. The crude was purified by passing
5
1
609.
. Chen, G.-M.; Brown, H. C. J. Am. Chem. Soc. 2002,
22, 4217.
6
7
1
through a short pad of silica gel to give the amine as
. (a) Buckett, W. R.; Thomas, P. C.; Luscomb, G. P.
1
colorless oil (387 mg, 87% yield). H NMR (CDCl ,): l
Prog. Neuropsychopharmacol. Biol. Psychiatry 1988, 12,
3
1
5
.80 (br S, 1H). 3.81 (S, 3H), 3.87 (S, 3H), 7.20–7.48 (m,
5
(
2
75; (b) Jerussi, T. P.; Senanayake, C. H.; Fang, Q. K.
Sepracor, Inc.) PCT Appl. 20020010198, 29 January,
001.
13
H), 7.50 (d, J=8.2 Hz, 2H), 7.60 (d, J=8.1 Hz, 2H);
C
NMR (CDCl ,): l 52.6, 53.2, 110.7, 119.1, 127.2, 128.2,
3
128.6, 128.7, 132.2, 139.9, 146.1; HRMS: calcd for
8
. Acyloxyborane species were reported previously, see:
Lane, C. F.; Myatt, H. L.; Daniels, J.; Hopps, H. B. J.
Org. Chem. 1974, 39, 3052; Brown, H. C.; Stocky, T.
P. J. Am. Chem. Soc. 1977, 99, 8218.
C H N (M+H) 223.1233, found 223.1235.
15
14
2
1
4. 4-(Benzylamino-methyl)-benzoic acid methyl ester was
prepared according to the general procedure in 92% yield
1
as colorless oil. H NMR (CDCl ,): l 1.70 (br S, 1H);
3
9
. For protocols of bifunctional catalysts, see: Sibi, M. P.;
Cook, G. R.; Liu, P. Tetrahedron Lett. 1999, 40, 2477;
Steinhagen, H.; Helmchen, G. Angew. Chem., Int. Ed.
Engl. 1996, 35, 2339 and references cited therein;
Fields, L. B.; Jacobsen, E. N. Tetrahedron: Asymmetry
3
5
.82 (S, 3H), 3.88 (S, 3H), 3.90 (S, 3H), 7.25–7.40 (m,
13
H), 7.50 (d, J=7.2 Hz, 2H), 8.0 (d, J=7.0 Hz, 2H);
C
NMR (CDCl ,): l 52.1, 52.8, 53.2, 126.4, 127.2, 128.1,
3
128.2, 128.5, 128.9, 129.8, 140.1, 145.8, 167.1; HRMS:
calcd for C H NO (M+H) 256.1326, found 256.1337.
16
17
2
1
993, 4, 2229; Ward, J.; Borner, A.; Kagan, H. B. Tet-
1
1
5. Benzyl-(4-styryl-benzyl) amine was prepared according to
rahedron: Asymmetry 1992, 3, 849; Borner, A.; Ward,
J.; Kortus, K.; Kagan, H. B. Tetrahedron: Asymmetry
the general procedure in 90% yield as colorless crystalline,
1
mp 42–44°C. H NMR (CDCl ,): l 1.70 (br S, 1H), 3.80
3
1
993, 4, 2219; Hola, J.; Quirmbach, M.; Borner, A.
13
(
S, 4H), 7.1–7.6 (m, 16H), C NMR (CDCl ,): l 52.9,
3
Synthesis 1997, 983; Williams, D. R.; Fromhold, M. G.
Synlett 1997, 523.
5
1
3.2, 126.6, 126.7, 127.1, 127.3, 127.6, 128.3, 128.4, 128.5,
28.6, 128.8, 136.1, 137.4, 139.9, 140.3; HRMS: calcd for
1
1
0. No reduction was observed with mandelic acid as the
additive. The readiness of reaction between borane and
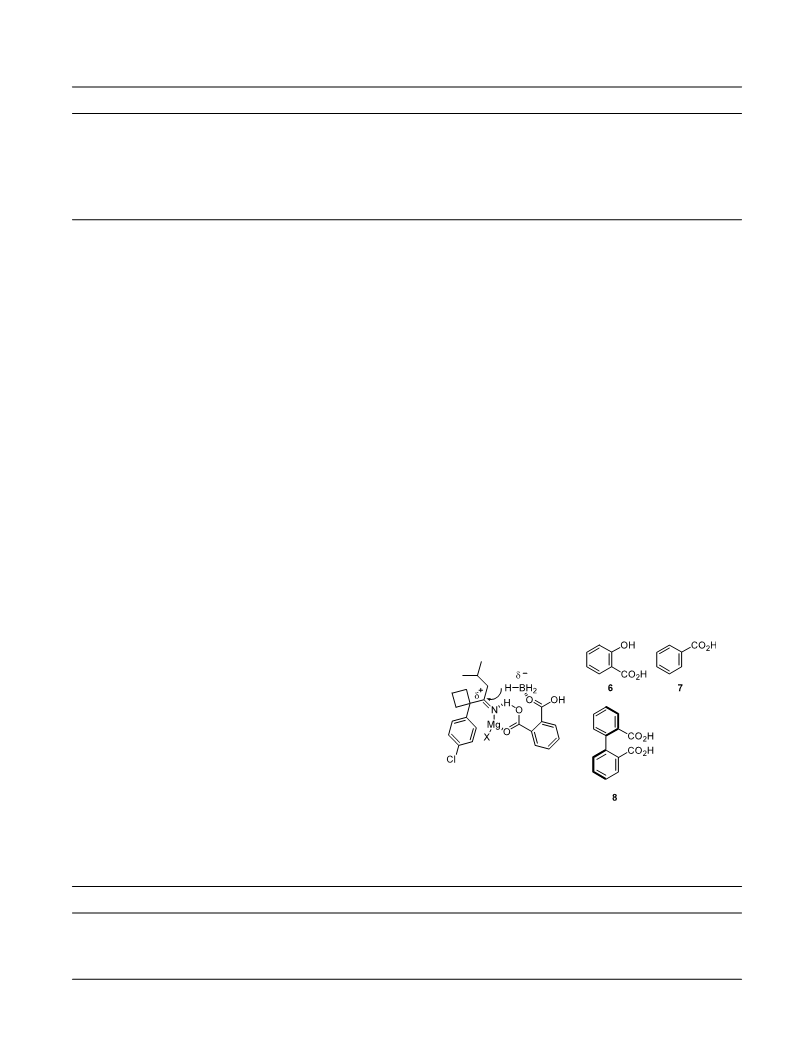
hydroxyl group to form alkoxyl borane species might
account for the inactivity of mandelic acid as catalyst.
1. For hydroboration of olefins, see: Brwon, H. C.;
Kramer, G. W.; Levy, A. B.; Midland, M. M. Organic
Synthesis via Boranes; John Wiley & Sons, 1975; Chap-
ter 1. For reduction of esters and nitriles, see: Brwon,
H. C.; Choi, Y. M.; Narasimhan, S. J. Org. Chem. 1982,
C H N (M+H) 300.1749, found 300.1752.
22
21
6. (1,3-Diphenyl-but-2-enyl)-phenylamine was prepared
according to the general procedure by adding BH ·THF
3
(
3.85 mL, 1 M in THF, 3.85 mmol) to a pre-mixed
solution of the imine (500 mg, 1.68 mmol) and phthalic
acid (639 mg, 3.85 mmol) in 10 mL THF at below −25°C.
Reaction completed in 6 min. The amine was obtained as
1
a white solid (480 mg, 95% yield). H NMR (CDCl ,): l
3
7.29–7.54 (m, 10H), 7.20 (t, J=7.5 Hz, 2H), 6.76 (t,
J=7.3 Hz, 1H), 6.66 (d, J=7.6 Hz, 2H), 6.01 (dd, J=8.5
Hz, J%=1.2 Hz, 1H), 5.33 (dd, J=8.49 Hz, J%=8.43 Hz,
1H), 4.15 (d, J=4.15 Hz, 1H), 2.26 (d, J=1.3 Hz, 3H);
4
7, 3153.
1
2. Preliminary experiment showed that treatment of the
imine 2 with 1 equiv. of (D)-ditoluoyl tartaric acid
13
(
DTTA) in THF at 22°C, followed by 1 equiv. of
C NMR (CDCl ,): l 147.56, 143.25, 142.76, 137.04,
3
borane at −60ꢀ−30°C, provided the amine 3 in 20%
ee.
130.52, 129.29, 128.93, 128.38, 127.45, 127.35, 126.87,
125.98, 117.73, 113.58, 57.10, 16.74; MS: 298.09 (M −1).
+

 Lu, Zhi-Hui
Lu, Zhi-Hui
 Bhongle, Nandkumar
Bhongle, Nandkumar
 Su, Xiping
Su, Xiping
 Ribe, Seth
Ribe, Seth
 Senanayake, Chris H
Senanayake, Chris H