T.J. Brown et al. / Journal of Organometallic Chemistry 758 (2014) 25e28
27
distances (Dd) of 2 relative to those of 4. Also worth noting is the
orientation of the coordinated C1]C2 bond in complexes 4, as
defined by the AueC1eC2 plane, relative to the P1 ligand, as
defined by AuePeC(ipso) plane, ranges from 12ꢁ in the case of 4a to
w70e75ꢁ in the case of 4c with 2 (35ꢁ) falling between these ex-
tremes. As we have previously noted in the context of gold p-alkene
complexes [15,20], the rotational orientation of the C]C bond
relative to the (L)Au fragment appears to be controlled primarily by
steric interactions without any notable electronic preference.
3. Conclusion
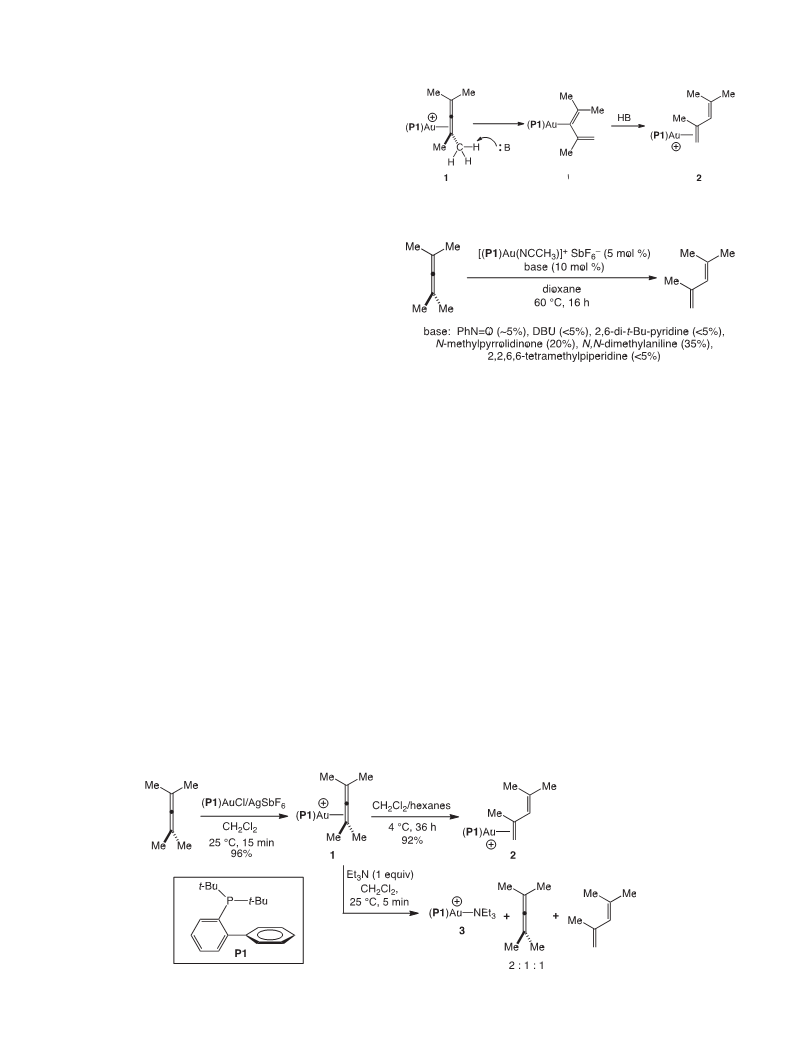
We have reported the isomerization of the cationic gold tetra-
methylallene complex [(P1)Au(h
2-Me2C]C]CMe2)]þ SbFe6 (1) via
formal 1,3-hydrogen migration to form the conjugated diene
complex {(P1)Au[
h
2-H2C]C(Me)ꢀC(H)]CMe2]}þ SbFꢀ6 (2), which
occurs at or below room temperature in the absence of any
apparent Brønsted base. These observations establish the ability of
cationic gold(I) complexes to mediate the conversion of aliphatic
Fig. 1. ORTEP drawing of 2ꢃCH2Cl2. Ellipsoids are shown at the 50% probability level
allenes to conjugated dienes and further reveals significant
acidity of the cationic, twelve electron (P1)Auþ fragment [15]. We
also report the X-ray crystal structure of gold -diene complex 2,
which was distinguished from related gold diene complexes 4 by
the more pronounced slippage of the coordinated C1]C2 bond due
to the C2 methyl group.
p-
with counterion, solvent, and hydrogen atoms omitted for clarity. Selected bond dis-
ꢀ
tances (A), bond angles (deg), and torsion angles (deg): AueC1 ¼ 2.202(4), Aue
p
C2
¼
2.390(4), C1eC2
¼
1.395(7), C2eC3
¼
1.458(7), C3eC4
¼
1.356(7), C2e
C5 ¼ 1.505(6), Au‒arene(plane) ¼ 3.04, AueC22 ¼ 3.08, PeAueC1]C2(cent) ¼ 160.2,
C1eC2eC3 ¼ 117.6(4), C1eC2eC3eC4 ¼ 175.8(5), C16eC21eC22eC23 ¼ 101.2(5), C16e
C21eC22eC27 ¼ 85.0(5).
4. Experimental
near planar s-trans configuration with a C1eC2eC3eC4 dihedral
angle of 175.8ꢁ. The diene ligand is positioned such that the plane
defined by AueC1eC2 is rotated w35ꢁ relative to the plane defined
by AuePeC16 with the C]CMe2 group directed away from the
protruding phenyl ring. The protruding phenyl group of the o-
biphenylphosphine moiety is nearly perpendicular to the P-bound
aryl ring with a C16eC21eC22eC27 dihedral angle of 85ꢁ. The
distance between the gold atom and the ipso carbon of the pro-
4.1. {[P(t-Bu)2o-biphenyl]Au[h
2-H2C]C(Me)C(H)]CMe2]}þ SbF6‒ (2)
Slow vapor diffusion of hexanes (15 mL) into a solution of 1
(62 mg, 7.5 ꢂ 10ꢀ2 mmol, 75 mM) in CH2Cl2 (1 mL) at 4 ꢁC for 36 h
formed colorless prismatic crystals of 2$CH2Cl2 which were sepa-
rated from the mother liquor, rinsed with cold hexanes (3 ꢂ 5 mL),
and dried to give 2$CH2Cl2 (57 mg, 92%). 1H NMR:
d 7.93e7.88 (m,
ꢀ
truding phenyl ring (C22) is 3.08 A, suggesting the presence of a
1 H), 7.65e7.55 (m, 5 H), 7.28e7.20 (m, 3 H), 5.84 (s, 1 H), 3.89 (d,
J ¼ 3.5 Hz, 1 H), 3.81 (d, J ¼ 4.0 Hz, 1 H), 2.27 (s, 3 H), 1.96 (s, 3 H),
weak Arearene interaction, as has been previously noted for
cationic gold complexes containing the P1 ligand [10e12,19].
Comparison of the structure of 2 to the structures of the related
1.92 (s, 3 H), 1.37 (d, J ¼ 16.5 Hz, 18 H). 31P NMR:
d 67.5. This spectral
data is consistent with the published spectroscopy of 2 [12].
gold
unsubstituted diene ligand provides insight into the effect of the
C2 methyl substituent of 2 on the gold -diene interaction (Table 1)
p-diene complexes 4 that contain a P1 ligand and a C1, C2-
4.2. In situ conversion of 1 to 2
p
[12]. The most notable difference between 2 and complexes 4 is the
more pronounced slippage of gold toward the terminal C1 atom of
the diene ligand. This slippage is evidenced both in the greater
deviation from linearity of the PeAueC]C(cent) angle of 2 relative
to 4 and in the greater difference between the AueC1 and AueC2
A solution of 1 (25 mg, 3.0 ꢂ 10ꢀ2 mmol, 5.5 ꢂ 10ꢀ2 mM) and
1,3-dimethoxybenzene (1.0
mL, 7.6 mmol; internal standard) in
CD2Cl2 (0.55 mL) was monitored periodically by 1H and 31P NMR
spectroscopy at 25 ꢁC. The relative concentrations of 1 and 2 were
determined by integrating the methyl resonance of 1 at
the olefinic resonances of 2 at
3.89 (d, J ¼ 3.5 Hz, 1H) and 3.81 (d,
J ¼ 4.0 Hz, 1H) relative to the methoxy resonance of 1,3-
dimethoxybenzene at
3.71 in the 1H NMR spectrum and by
integrating the phosphorous resonances of 1 ( 66.7) and 2 ( 67.5)
d 1.93 and
d
Table 1
Comparison of geometric parameters defining the slippage and rotational orienta-
tion of the diene ligand of complexes 2$CH2Cl2, 4a, 4b, and 4c.
d
d
d
in the 31P NMR spectrum. in situ Analysis of the conversion of 1
(55 mM in CD2Cl2) to 2 in the presence of 2,4-dimethyl-2,3-
pentadiene (55 mM) and the reaction of 1 (55 mM in CD2Cl2)
with triethylamine (55 mM) were performed employing similar
procedures.
4.3. X-ray data crystal structure of 2$CH2Cl2
Compound
Dd AueC1/AueC2 PeAue(C1]C2)cent (AuePeCipso)e(AueC1eC2)
ꢀ
(A)
(deg)
(deg)
A crystal of 2$CH2Cl2 was mounted on a Mitegen polyimide
micromount with a small amount of Paratone N oil and analyzed on
a Bruker-Nonius Kappa Axis X8 Apex2 diffractometer at 110 K. The
unit cell dimensions were determined from a symmetry con-
2
0.19
0.09
0.11
0.08
160
167
169
165.5
35
20
13
72
4aa,b
4ba,b
4ca
strained fit of 9966 reflections with 4.36ꢁ < 2
q
< 57.98ꢁ. The data
u and 4 scans, collected up to
a
Taken from reference [12].
Average of two disordered molecules.
b
collection strategy was a number of

 Brown, Timothy J.
Brown, Timothy J.
 Robertson, Bradley D.
Robertson, Bradley D.
 Widenhoefer, Ross A.
Widenhoefer, Ross A.