A Rapid, Efficient Method for Deprotection of Oximes
Letters in Organic Chemistry, 2015, Vol. 12, No. 1 75
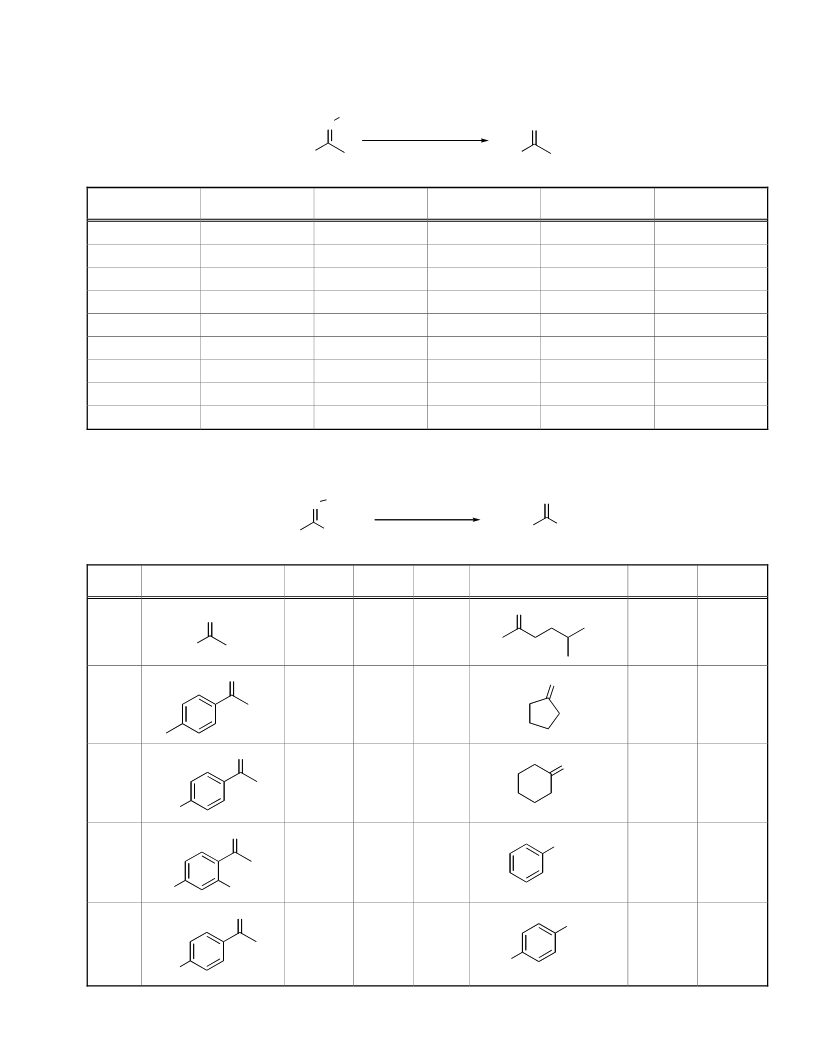
Table 2. Contd…….
b
b
Entry
Product
Time/min
Yield
Entry
Product
Time/min
Yield
CHO
CHO
e
6
8
92
13
5
83
Cl
MeO
2
f
2m
O
f
7
8
91
14
5
76
CHO
2
n
2
g
a,Reaction condition: oxime (2 mmol), NaClO
2
(2 mmol), concentrate HCl (2mL) , H
2
O (2 mL), room temperature. b, Isolated yield. c, the isolated yield of acid is 9%. d, the isolated
yield of acid is 8%. e, the isolated yield of acid is 7%. f, the isolated yield of acid is 8%.
thin-layer chromatography, TLC). The reaction was ex-
tracted with ethyl acetate (10 mL). The combined organic
extract was washed with water (5 mL) and dried with anhy-
REFERENCES
[1]
Sandler, S. R.; Karo, W. Organic functional group preparations,
2nd ed., vol. 3; Academic Press: San Diego, 1989.
[2]
Ghiaci, M.; Hassan, I. G. A facile beckmann rearrangement of
drous MgSO
silica gel column (200–300 mesh) to afford the product 2a
0.22 g, 92%). The carbonyl compounds formed were char-
4
. Then it was directly filtered through a short
oximes with AlCl
275-2280.
3
in the solid state Synth. Commun., 1998, 28,
2
(
[3]
Peter W.; Joan M. F.; Laura S., Microwave promoted oxazole
synthesis: cyclocondensation cascade of oximes and acyl chlorides
Tetrahedron Lett., 2005, 46, 5463-5466.
acterized by their physical data, which were in accordance
with values reported in the literature, and GC-MS analysis.
[
4]
5]
Karipcin, F.; Karatas, ꢁ.; Ucan, H. ꢁ. Some polyamidoxime deriva-
tives and their metal complexes Turk. J. Chem., 2003, 27, 453-460.
Zhang, G. S.; Gong, H.; Yang, D. H.; Chen, M. F. The Cleavage of
p-nitrophenylhydrazones and semicarbazones with ammonium
chlorochromate adsorbed on alumina under non-aqueous conditions
Synth. Commun. 1999, 29, 1165-1170.
1
HNMR of some typical products is given below:
[
1
Acetophenone (2a). H NMR (500 MHz, CDCl
3
): ꢀ 2.57
(
s, 3H), 7.39 - 7.56 (m, 3H), 7.90 - 7.96 (m, 2H).
1
p-Methylacetophenone (2b). H NMR (500 MHz,
[6]
Kurangi, R. F.; Kawthankar, R.; Sawal, S.; Desai, V. G.; Tilve, S.
G. Convenient synthesis of 3,5-disubstituted isoxazoles Synth.
Commun., 2007, 37, 585-587.
CDCl
3
): ꢀ 2.38 (s, 3H), 2.54 (d, 3H), 7.22 (d, J= 5.96 Hz,
2
H), 7.83 (d , J= 5.96 Hz, 2H).
[7]
[8]
[9]
Liu, J.; Li, D. F.; Li, J. A.; Li, C. J.; Jia, X. S. Regioselective syn-
thesis of functionalized oxime ethers Lett. Org. Chem. 2010, 7,
1
3
p-Nitroacetophenone (2e). H NMR (500 MHz, CDCl ):
4
79-482.
ꢀ
2.67 (s, 3H), 8.11 (d, J= 6.96 Hz, 2H), 8.28 (d , J= 7.08 Hz,
Greene, T. W.; Wuts, P. G. M. In: Protective groups in organic
synthesis, 4th ed.; John Wiley: New York, NY, USA, 2007, pp.
2
H).
5
06–527.
1
5
-Methyl-2-hexanone (2h). H NMR (500 MHz, CDCl
3
):
Kabalka, G. W.; Pace, R. D.; Wadgonkar, P. P. The palladium
assisted transfer reduction of ꢂ, ꢃ-unsaturated nitroalkenes to oxi-
mes using ammonium formate Synth. Commun., 1990, 20, 2453-
ꢀ
2
8
0.81-0.83 (m, 6H), 1.37-1.42 (m, 2H), 1.44-1.49 (m, 1H),
.06 ( d, 3H), 2.34-2.37 (m, 2H).8.11 (d, J= 6.96 Hz, 2H),
.28 (d , J= 7.08 Hz, 2H).
2
458.
[10]
Shinada, T.; Yoshihara, K. A facile method for the conversion of
1
2
oximes to ketones and aldehydes by the use of activated MnO Tet-
rahedron Lett., 1995, 36, 6701-6704.
Cyclopentanone (2i). H NMR (500 MHz, CDCl
3
): ꢀ
1
.91-1.94 (m, 4H), 2.11-2.14 (m, 4H).
[
11]
12]
Zhao, W. C.; Sha, Y. W. Review of deoximation methods Chin. J.
Org. Chem., 1996, 16, 121-132.
Shriner, R. L.; Fuson, R. C.; Curtin, D. Y.; Morrill, T. C. The sys-
tematic identification of organic compounds, 6th ed.; John Wiley:
New York, 1980.
1
Benzaldehyde (2k). H NMR (500 MHz, CDCl
3
): ꢀ 7.50-
[
7
1
.53 (m, 2H), 7.63-7.65 (m, 1H), 7.86-7.88 (m,2H), 10.01 (s,
H).
1
[13]
Wang, K.; Qian, X.; Cui, J. T. One step from nitro to oxime: a
convenient preparation of unsaturated oximes by the reduction of
the corresponding vinylnitro compounds Tetrahedron 2009, 65,
10377-10382.
Domingo, L. R.; Picher, M. T.; Arroyo, P.; Sez, J. A. 1, 3-Dipolar
cycloadditions of electrophilically activated benzonitrile N-oxides.
polar cycloaddition versus oxime formation J. Org. Chem. 2006,
Isobutyraldehyde (2n). H NMR (500 MHz, CDCl
3
): ꢀ
1
1
.10 (d, J= 5 Hz, 6H), 2.39-2.42 (m, 1H), 9.62 (d, J= 1 Hz,
H)
[14]
CONFLICT OF INTEREST
7
1, 9319-9330.
[
15]
16]
Czekelius, C.; Carreira, E. M. Convenient transformation of opti-
cally active nitroalkanes into chiral aldoximes and nitriles Angew.
Chem. Int. Ed. 2005, 44, 612-615.
Quan, N.; Shi, X. X.; Nie, L. D.; Dong, J.; Zhu, R. H. A green
chemistry method for the regeneration of carbonyl compounds
from oximes by using cupric chloride dihydrate as a recoverable
promoter for hydrolysis Synlett 2011, 1028-1032.
Majireck, M. M.; Witek, J. A.; Weinreb, S. M. An expedient reduc-
tive method for conversion of ketoximes to the corresponding car-
bonyl compounds Tetrahedron Lett. 2010, 51, 3555-3557.
The authors confirm that this article content has no con-
flict of interest.
[
ACKNOWLEDGEMENTS
We gratefully acknowledge financial support by the
National Natural Science Foundation of China (No.
[
17]
2
1242004, No. 21302168).

 Yao, Yuan-Yuan
Yao, Yuan-Yuan
 Zhao, Xian-Liang
Zhao, Xian-Liang