Related Products
- 93549-15-6Isoquinoline,3,4-dihydro-6-methoxy-, hydrochloride (1:1)
- 93-55-01-Propanone,1-phenyl-
- 935521-01-03,4-Quinolinediamine;N4-(2-methylpropyl)-,monohydrochloride
- 935525-13-6BMS 433796
- 935534-47-75-bromo-4-(trifluoromethyl)pyrimidin-2-amine
- 93553-81-2Butanedioic acid,2-(3,4-dichlorophenyl)-
- 93554-80-42-aminobutanenitrile
- 935-56-81-Chloroadamantane
- 93-56-1Styrene glycol
- 935661-15-71H-Indazole,4-iodo-1-methyl-
Hot Products
- 104987-11-3Tacrolimus
- 141-53-7Sodium formate
- 8001-54-5Quaternary ammonium compounds, alkylbenzyldimethyl, chlorides
- 9003-39-8Povidone
- 10161-34-9Trenbolone acetate
- 402957-28-2Telaprevir
- 68-19-9Cyanocobalamin
- 7631-86-9Silicon dioxide
- 302-79-4Tretinoin
- 77-92-9Citric acid

|
Basic Information |

|
Post buying leads |

|
Suppliers |

|
Cas Database |

| Name |
1-Phenyl-1-propanol |
EINECS | 202-256-0 |
| CAS No. | 93-54-9 | Density | 0.994 g/cm3 |
| PSA | 20.23000 | LogP | 2.13000 |
| Solubility | insoluble in water | Melting Point |
N/A |
| Formula | C9H12O | Boiling Point | 219 °C at 760 mmHg |
| Molecular Weight | 136.194 | Flash Point | 97.3 °C |
| Transport Information | UN 2810 | Appearance | clear colorless liquid |
| Safety | 23-24/25 | Risk Codes | 22 |
| Molecular Structure |
|
Hazard Symbols |
 Xn Xn
|
| Synonyms |
Benzylalcohol, a-ethyl- (8CI);(RS)-1-Phenylpropan-1-ol;(RS)-1-Phenylpropanol;1-Hydroxy-1-phenylpropane;1-Phenyl-1-hydroxypropane;1-Phenyl-n-propanol;1-Propanol, 1-phenyl-;Bilergon;Carbicol;Ejibil;Epatoxfen;Felicur;Felitrope;Fepar;Livonal;NSC 41708;Phenicol;Phenycholon;Phenylchol;SH 261;a-Ethylbenzenemethanol;a-Hydroxypropylbenzene; |
Article Data | 544 |
1-Phenyl-1-propanol Synthetic route
Conditions

| Conditions | Yield |
|---|---|
| With methylaluminum bis(2,6-di-tert-butylphenoxide) In diethyl ether; toluene at -78℃; for 2h; | 100% |
| In diethyl ether at 0 - 20℃; | 87% |
| Stage #1: ethylmagnesium bromide; benzaldehyde In tetrahydrofuran at 30℃; for 24h; Inert atmosphere; Stage #2: With water In tetrahydrofuran Inert atmosphere; | 73% |
Conditions

| Conditions | Yield |
|---|---|
| With potassium hydride; ditriptylethylborane In tetrahydrofuran for 0.0833333h; | 100% |
| With zirconium dioxide hydrate; isopropyl alcohol at 130℃; for 0.5h; Meerwein-Ponndorf-Verley Reduction; | 100% |
| With hydrogen In methanol at 40℃; for 24h; chemoselective reaction; | 100% |
Conditions

| Conditions | Yield |
|---|---|
| With C35H41ClFeIrPS; hydrogen; sodium methylate In tetrahydrofuran at 25℃; under 22502.3 Torr; for 2h; Reagent/catalyst; Autoclave; Inert atmosphere; | 100% |
| With sodium tetrahydroborate; C11H18Cl2CoN2S; hydrogen In isopropyl alcohol at 100℃; under 37503.8 Torr; for 16h; Glovebox; Autoclave; | 97% |
| With 1,1'-bis(diphenylphosphino)ferrocene; [ruthenium(II)(η6-1-methyl-4-isopropyl-benzene)(chloride)(μ-chloride)]2; Butane-1,4-diol; potassium tert-butylate at 110℃; for 24h; Inert atmosphere; | 92% |
- 321884-15-5
2-(1-phenyl-propoxy)-tetrahydro-pyran

- 93-54-9
1-Phenyl-1-propanol
Conditions

| Conditions | Yield |
|---|---|
| silica-supported prop-1-ylsulfonic acid In methanol | 99.6% |
| With tetra-N-butylammonium tribromide In methanol at 20℃; for 1h; | 97% |
| With tetra-N-butylammonium tribromide In methanol for 1.2h; | 97% |
| With trichloroisocyanuric acid In methanol at 20℃; for 4h; | 92% |
Conditions

| Conditions | Yield |
|---|---|
| With (1R,2S)-2-aminomethyl-2-hydroxy-1,7,7-trimethylbicyclo[2.2.1]heptane In hexane for 48h; | 99% |
| With polystyrene bond triphenylphosphonium chloride In toluene at 0 - 23℃; Inert atmosphere; chemoselective reaction; | 99% |
| With 15-crown-5; sodium iodide In toluene at 0 - 23℃; for 24h; Reagent/catalyst; Solvent; Inert atmosphere; Schlenk technique; | 99% |
Conditions

| Conditions | Yield |
|---|---|
| With hydrogen; TMSB; palladium In ethanol at 20℃; under 760 Torr; for 20h; | 98% |
| With riboflavin tetrabutyrate; oxygen; hydrazine hydrate In acetonitrile at 30℃; under 760.051 Torr; for 30h; | 92% |
| With oxygen; hydrazine hydrate; riboflavin 2’,3’,4’,5’-tetrabutanoate In acetonitrile at 25 - 30℃; for 30h; chemoselective reaction; | 92% |
Conditions

| Conditions | Yield |
|---|---|
| With titanium tetrachloride In dichloromethane at -78 - -30℃; Product distribution; further aldehydes and tetraalkylleads; reaction with mixed tetraalkylleads; other conditions; | 98% |
| With titanium tetrachloride In dichloromethane at -78 - -30℃; | 98% |
| With titanium tetrachloride In dichloromethane at -78 - -30℃; | 96% |
- 64346-32-3
triethyl-butyl plumbane

- 100-52-7
benzaldehyde

A
- 93-54-9
1-Phenyl-1-propanol

B
- 583-03-9
1-Phenyl-1-pentanol
Conditions

| Conditions | Yield |
|---|---|
| With titanium tetrachloride In dichloromethane at -78 - -30℃; | A 98% B 1.7% |
- 136116-42-2
1-phenyl-1-tert-butyldimethylsiloxypropane

- 93-54-9
1-Phenyl-1-propanol
Conditions

| Conditions | Yield |
|---|---|
| With tetra-N-butylammonium tribromide In methanol for 7h; | 98% |
| With N-iodo-succinimide In methanol at 20℃; for 19h; | 95% |
| With methanol at 20℃; for 3.33333h; | 93% |
| sulfonic acid functionalized nanoporous silica In methanol at 35℃; for 12h; | 87% |
| With tetrabutyl ammonium fluoride |
Conditions

| Conditions | Yield |
|---|---|
| In toluene for 0.166667h; Alkylation; reduction; Title compound not separated from byproducts.; | A 97% B 2% |
1-Phenyl-1-propanol Chemical Properties
Molecular Formula:C9H12O
Molar mass:136.19098g/mol
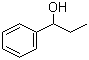
Structure of 1-Phenyl-1-propanol(93-54-9):
Synonyms of 1-Phenyl-1-propanol(93-54-9):.alpha.-Hydroxypropylbenzene;alpha-ethylbenzyl alcohol;1-Phenyl-1-propanol;Gallenperlen;Phenycholon;1-Phenyl-1-Hydroxypropane;1-Phenylpropan-1-ol;1-phenyl-propan-1-ol;1-Propanol, 1-phenyl-
Density:0.994 g/cm3
Flash Point:97.3 °C
Boiling Point:219 °C at 760 mmHg
Index of Refraction:1.524
Vapour Pressure:0.0707 mmHg at 25°C
Water solubility:insoluble
Appearance:Colorless oily liquid
Molar mass:136.19098g/mol
Structure of 1-Phenyl-1-propanol(93-54-9):

Synonyms of 1-Phenyl-1-propanol(93-54-9):.alpha.-Hydroxypropylbenzene;alpha-ethylbenzyl alcohol;1-Phenyl-1-propanol;Gallenperlen;Phenycholon;1-Phenyl-1-Hydroxypropane;1-Phenylpropan-1-ol;1-phenyl-propan-1-ol;1-Propanol, 1-phenyl-
Density:0.994 g/cm3
Flash Point:97.3 °C
Boiling Point:219 °C at 760 mmHg
Index of Refraction:1.524
Vapour Pressure:0.0707 mmHg at 25°C
Water solubility:insoluble
Appearance:Colorless oily liquid
1-Phenyl-1-propanol Uses
1-Phenyl-1-propanol(93-54-9) is also a gallbladder medicine,and can be used as a spice and heat-transfering medium.
1-Phenyl-1-propanol Production
Derived from the reduction of acetone from benzene in ethanol by potassium borohydride .
1-Phenyl-1-propanol Toxicity Data With Reference
| 1. | orl-rat LD50:1600 mg/kg | ARZNAD Arzneimittel-Forschung. Drug Research. 12 (1962),347. | ||
| 2. | orl-mus LD50:500 mg/kg | AIPTAK Archives Internationales de Pharmacodynamie et de Therapie. 116 (1958),154. | ||
| 3. | scu-mus LD50:700 mg/kg | AIPTAK Archives Internationales de Pharmacodynamie et de Therapie. 116 (1958),154. |
Other: See actual entry in RTECS for complete information.The toxicological properties have not been fully investigated. Mutagenicity: Ames-test: negative.
1-Phenyl-1-propanol Safety Profile
Moderately toxic by ingestion and subcutaneous routes. When heated to decomposition it emits acrid smoke and irritating fumes.
Hazard Codes:Xn
Risk Statements:
22: Harmful if swallowed
Safety Statements:
23: Do not breathe gas/fumes/vapor/spray (appropriate wording to be specified by the manufacturer)
24: Avoid contact with skin
25: Avoid contact with eyes
Hazard Codes:Xn

Risk Statements:
22: Harmful if swallowed
Safety Statements:
23: Do not breathe gas/fumes/vapor/spray (appropriate wording to be specified by the manufacturer)
24: Avoid contact with skin
25: Avoid contact with eyes
1-Phenyl-1-propanol Specification
Stability and Reactivity of 1-Phenyl-1-propanol(93-54-9):
Conditions to Avoid:Incompatible materials.
Chemical Stability:Stable under normal temperatures and pressures.
Incompatibilities with Other Materials:Incompatible materials, acid anhydrides, acid chlorides, acids.
Hazardous Decomposition Products: carbon dioxide,Carbon monoxide.
Conditions to Avoid:Incompatible materials.
Chemical Stability:Stable under normal temperatures and pressures.
Incompatibilities with Other Materials:Incompatible materials, acid anhydrides, acid chlorides, acids.
Hazardous Decomposition Products: carbon dioxide,Carbon monoxide.

