717906-29-1 Usage
Description
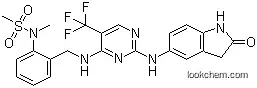
N-Methyl-N-[2-[[[2-[(2-oxo-2,3-dihydro-1H-indol-5-yl)amino]-5-trifluoromethylpyrimidin-4-yl]amino]methyl]phenyl]methanesulfonamide, also known as PF-431396, is a pyrimidine-based dual inhibitor of proline-rich tyrosine kinase 2 (PYK2) and focal adhesion kinase (FAK) with IC50 values of 2 and 11 nM, respectively. It is a compound that has been developed to target specific nonreceptor tyrosine kinases, which play a role in various cellular processes, including cell adhesion, migration, and proliferation.
Uses
1. Used in Pharmaceutical Industry:
N-Methyl-N-[2-[[[2-[(2-oxo-2,3-dihydro-1H-indol-5-yl)amino]-5-trifluoromethylpyrimidin-4-yl]amino]methyl]phenyl]methanesulfonamide is used as a dual PYK2/FAK inhibitor for studying its effects on various cellular processes and signaling pathways. It has been shown to promote osteoblast recruitment and activity, stimulating bone formation in ovariectomized rats, which could be beneficial for the development of treatments for osteoporosis and other bone-related conditions.
2. Used in Research Applications:
N-Methyl-N-[2-[[[2-[(2-oxo-2,3-dihydro-1H-indol-5-yl)amino]-5-trifluoromethylpyrimidin-4-yl]amino]methyl]phenyl]methanesulfonamide is used as a research tool to study the roles of PYK2 and FAK in various cellular processes, such as cell adhesion, migration, and proliferation. It has been used to investigate the effects of PYK2/FAK inhibition on phosphorylation of Yes-associated protein (YAP) at Ser127 and the steady state of transcriptional coactivator with PDZ-binding motif (TAZ). Additionally, it has been utilized to study the effects of PYK2/FAK inhibition on protein kinase A activation in human sperm samples, which could provide insights into male fertility and reproductive health.
Biochem/physiol Actions
PF-431396 is a potent inhibitor of PYK2 and FAK kinases (IC50 = 11 and 1.5 nM, respectively). PF-431396 increases bone formation and protects against bone loss in ovariectomized rats.
Check Digit Verification of cas no
The CAS Registry Mumber 717906-29-1 includes 9 digits separated into 3 groups by hyphens. The first part of the number,starting from the left, has 6 digits, 7,1,7,9,0 and 6 respectively; the second part has 2 digits, 2 and 9 respectively.
Calculate Digit Verification of CAS Registry Number 717906-29:
(8*7)+(7*1)+(6*7)+(5*9)+(4*0)+(3*6)+(2*2)+(1*9)=181
181 % 10 = 1
So 717906-29-1 is a valid CAS Registry Number.
InChI:InChI=1S/C22H21F3N6O3S/c1-31(35(2,33)34)18-6-4-3-5-13(18)11-26-20-16(22(23,24)25)12-27-21(30-20)28-15-7-8-17-14(9-15)10-19(32)29-17/h3-9,12H,10-11H2,1-2H3,(H,29,32)(H2,26,27,28,30)
717906-29-1Relevant articles and documents
Trifluoromethylpyrimidine-based inhibitors of proline-rich tyrosine kinase 2 (PYK2): Structure-activity relationships and strategies for the elimination of reactive metabolite formation
Walker, Daniel P.,Christopher Bi,Kalgutkar, Amit S.,Bauman, Jonathan N.,Zhao, Sabrina X.,Soglia, John R.,Aspnes, Gary E.,Kung, Daniel W.,Klug-McLeod, Jacquelyn,Zawistoski, Michael P.,McGlynn, Molly A.,Oliver, Robert,Dunn, Matthew,Li, Jian-Cheng,Richter, Daniel T.,Cooper, Beth A.,Kath, John C.,Hulford, Catherine A.,Autry, Christopher L.,Luzzio, Michael J.,Ung, Ethan J.,Roberts, W. Gregory,Bonnette, Peter C.,Buckbinder, Leonard,Mistry, Anil,Griffor, Matthew C.,Han, Seungil,Guzman-Perez, Angel
scheme or table, p. 6071 - 6077 (2009/08/07)
The synthesis and SAR for a series of diaminopyrimidines as PYK2 inhibitors are described. Using a combination of library and traditional medicinal chemistry techniques, a FAK-selective chemical series was transformed into compounds possessing good PYK2 potency and 10- to 20-fold selectivity against FAK. Subsequent studies found that the majority of the compounds were positive in a reactive metabolite assay, an indicator for potential toxicological liabilities. Based on the proposed mechanism for bioactivation, as well as a combination of structure-based drug design and traditional medicinal chemistry techniques, a follow-up series of PYK2 inhibitors was identified that maintained PYK2 potency, FAK selectivity and HLM stability, yet were negative in the RM assay.