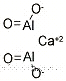12042-68-1 Usage
Description
Calcium aluminate, also known as CaAl2O4, is a white powder with a monoclinic crystalline form. It has a density of 2.98 g/cc and a melting point of 1605 °C. It is commonly referred to as "aluminous cement," "high-alumina cement" (HAC), or "Ciment fondu."
Uses
Used in Cement Industry:
Calcium aluminate is used as a hydrous cement for small-scale, specialized applications. It is known for its rapid hardening and high early strength properties, making it suitable for various construction projects.
Used in Catalyst Production:
Calcium aluminum oxide is used as a precursor in the production of various catalysts, including naphtha steam reforming catalysts, refinery gas steam reforming catalysts, and secondary hydrocarbon steam reforming catalysts. These catalysts play a crucial role in the petrochemical and refining industries.
Used in Refractory and Cement Production:
Calcium aluminate is also utilized in the production of refractory materials and cements due to its high-temperature stability and chemical resistance.
Physical and Chemical Properties:
Calcium aluminate exhibits a white, monoclinic, triclinic, or rhombic crystalline structure. It is available in a fine powder form, with -200 mesh 99% powder(s) being a common specification.
Preparation
Calcium aluminate can be formed by heating the
oxides together at about 900–1250 °C:
CaO + Al2O3 + heat→CaAl2O4
Check Digit Verification of cas no
The CAS Registry Mumber 12042-68-1 includes 8 digits separated into 3 groups by hyphens. The first part of the number,starting from the left, has 5 digits, 1,2,0,4 and 2 respectively; the second part has 2 digits, 6 and 8 respectively.
Calculate Digit Verification of CAS Registry Number 12042-68:
(7*1)+(6*2)+(5*0)+(4*4)+(3*2)+(2*6)+(1*8)=61
61 % 10 = 1
So 12042-68-1 is a valid CAS Registry Number.
InChI:InChI=1/2Al.Ca.4O/q;;+2;;;2*-1/r2AlO2.Ca/c2*2-1-3;/q2*-1;+2
12042-68-1Relevant articles and documents
Selective self-redox and crystal field modulation for enhanced and tuned broadband emission in chromium-doped aluminate glasses
Li, Xingyu,Luo, Haoyang,Wang, Yafei,Wang, Xiu,Cao, Jiangkun,Wang, Liping,Peng, Mingying
, p. 5401 - 5409 (2019)
The emerging era of big data has stimulated the development of optical materials with broad emission region to allow data transmission of higher capacity and speed. Here, broadband emission, almost covering the region of 650-1500 nm, was realized by doping transition metal ions into versatile aluminate glasses, in which abundant microstructures and certain oxidizing capacity could be guaranteed to enrich emissions. Furthermore, we aimed to improve spectroscopic properties via the simultaneous self-redox and crystal field modulation of Cr ions and matrixes, respectively. More emission ions and more efficient centers were created to enhance the emission intensity by more than one order of magnitude. In addition, proper crystal field was created to liberate a new emission band at ~708 nm. The results may allow the development of tunable fiber laser and amplifier using the materials and pave the way to precisely control the valence and crystal field to obtain efficient emission.
A mass-spectrometric study of vapor composition and thermodynamic properties of CaO-Al2O3 melts
Shornikov,Stolyarova,Shul'ts
, p. 19 - 22 (2007/10/03)
The composition and partial pressures of molecular species in vapor over melts of CaO-Al2O3 at 1833-2033 K were determined by Knudsen effusion-mass spectrometry. Negative deviations from ideality were observed for the system. The thermodynamic properties of CaO-Al2O3 melts were expressed by analytical dependences as functions of the temperature and concentration of components of the system.