Evaluation Only. Created with Aspose.PDF. Copyright 2002-2021 Aspose Pty Ltd.
Table 1 Yield of propan-2-ol and alkylphenols with water density for 60
The dehydration of propan-2-ol yielded propylene at the early
reaction times. Alkylphenols dissociate in its aqueous solution,
as shown by Xiang et al.11 who reported that b-naphthol
dissociates even in supercritical water. For example, the
dissociation constant of b-naphthol is 1.26 3 10211 mol kg21 at
673 K and 0.5 g cm23 of water density. At room temperature
and pressure, the dissociation constant of phenol (pKa = 9.89)
at 293 K and 0.1 MPa was similar to that of b-naphthol (pKa =
9.51) at 298 K and 0.1 MPa.12 Phenol probably dissociates in
supercritical water. Antal et al.13 reported that the acid-
catalyzed dehydration of propan-2-ol yielded propylene in high
temperature water at 593 K.
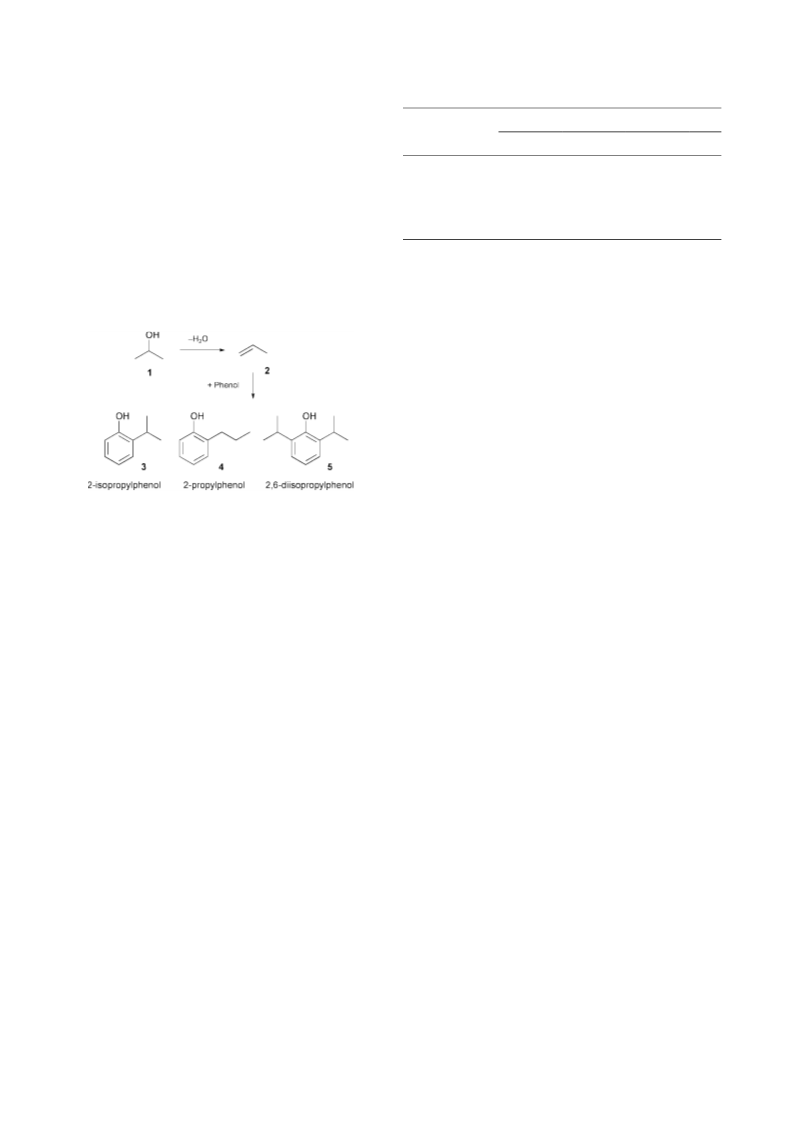
Considering these experimental findings, the reaction path-
way shown in Scheme 1 was developed. The dehydration of
propan-2-ol (1) probably yields propylene (2), following
alkylation of phenol with propylene to 2-isopropylphenol (3),
2-propylphenol (4) and 2,6-diisopropylphenol (5). It is probable
that phenol acts as an acid catalyst and promotes the
dehydration of propan-2-ol at supercritical conditions.
minutes of reaction time at 673 K, [propan-2-ol]0
[phenol]0 = 1.65 mol L21
=
0.33 mol L21
,
Yield (%)
Water density/
g cm23
1
3
4
5
0.3
0.4
0.42
0.46
0.48
0.5
75.9
58.5
13.7
4.5
3.6
2.6
3.1
0.3
2.2
5.8
7.1
7.5
6.9
0
18.0
45.5
55.1
58.8
55.0
0.6
3.7
5.1
5.4
5.1
water density. We consider that phase behavior and the
concentration of protons can affect the reaction. At low water
density, the reactants were separated into two phases. Phenol
would probably be mainly distributed in the liquid phase while
the propan-2-ol would be distributed in the gas phase. With
increasing water density, the phase in the reactor probably
became homogeneous. In addition, the dissociation constant of
b-naphthol increases with increasing water density in super-
critical water.11 This trend seems to be applicable to the
dissociation of phenol. If the dissociation constant of phenol
increased with increasing water density, the concentration of
protons also increased. Then, dehydration and alkylation was
promoted with increasing water density, because the proton
catalyzed both reactions.
In summary, phenol was alkylated with propan-2-ol without
catalyst in supercritical water at 673 K. The alkylphenols
obtained were mainly ortho substituted compounds and the
maximum yield of 2-isopropylphenol was 58.8%. The sum of
ortho alkylphenols yield was 71.7%. Further, alkylation was
enhanced with increasing water density. Our results show that
supercritical water can provide a unique reaction field for the
alkylation, which is non-catalytic and highly selective.
The authors thank a Grant-in-Aid for Scientific Research on
Priority Areas (09450281, 10555270, 11450295 and 11694921)
the Ministry of Education, Culture, Sports, Science and
Technology, for support of this research.
Scheme 1
The alkylation of phenol with propylene yielded 2-iso-
propylphenol, 2-propylphenol and 2,6-diisopropylphenol and
this reaction was the rate-determining step. Phenoxyether was
not produced, probably because hydrolysis of ethers occurs
readily in supercritical water.5 Phenol was alkylated almost
solely at the ortho position of the hydroxy group even in the
homogeneous system as obtained in high temperature water by
Chandler et al.7 The hydroxy group of phenol and several water
molecules can construct a ring structure formed by hydrogen-
bonds14 and the distance between hydroxy oxygen and
hydrogen increases with increasing the number of water
molecules in a ring structure.15 In supercritical carbon dioxide,
specific solvation around the hydroxy group of phenol can
occur.16 Taking these results into account, water molecules
probably locate around the hydroxy group of phenol at
supercritical conditions and construct a ring structure with the
hydroxy group of phenol. Then, the dissociation of the hydroxy
group is promoted locally around it. This makes the reaction
field limited only around the hydroxy group, which results in
mainly the ortho position being alkylated.
Notes and references
1 G. A. Olah, Friedel–Crafts and related reactions, Interscience Pub-
lishers, New York, 1963, vol I.
2 F. J. Sowa, G. F. Hennion and J. A. Nieuwland, J. Am. Chem. Soc., 1935,
57, 709.
3 S. Sato, R. Takahashi, T. Sodesawa, K. Matsumoto and Y. Kamimura,
J. Catal., 1999, 184, 180.
4 W. K. Gray, F. R. Smail, M. G. Hitzler, S. K. Ross and M. Poliakoff, J.
Am. Chem. Soc., 1999, 121, 10711.
5 P. E. Savage, Chem. Rev., 1999, 99, 603.
6 M. Siskin and A. R. Katritzky, J. Anal. Appl. Pyrolysis, 2000, 54,
193.
7 K. Chandler, F. Deng, A. K. Dillow, C. L. Liotta and C. A. Eckert, Ind.
Eng. Chem. Res., 1997, 36, 5175.
8 K. Chandler, C. L. Liotta, C. A. Eckert and D. Schiraldi, AIChE J., 1998,
44, 2080.
9 M. Sasaki, Z. Fang, Y. Fukushima, T. Adschiri and K. Arai, Ind. Eng.
Chem. Res., 2000, 39, 2883.
10 P. E. Savage, S. Gopalan, T. I. Mizan, C. J. Martino and E. E. Brock,
AIChE J., 1995, 41, 1723.
11 T. Xiang and K. P. Johnston, J. Phys. Chem., 1994, 98, 7915.
12 D. R. Lide, CRC Handbook of Chemistry and Physics 73rd Edition,
CRC Press, Inc., Boca Raton, 1992–1993.
13 M. J. Antal, M. Carlsson, X. Xu and D. G. M. Anderson, Ind. Eng.
Chem. Res., 1998, 37, 3820.
14 N. Mikami, Bull. Chem. Soc. Jpn., 1995, 68, 683.
15 Y. Dimotrova, J. Mol. Struct. (THEOCHEM), 1998, 455, 9.
16 N. Wada, M. Saito, D. Kitada, R. L. Smith, H. Inomata, K. Arai and S.
Saito, J. Phys. Chem. B, 1997, 101, 10918.
The data reported above were measured at 0.5 g cm23 of
water density. At these conditions, the phase in the reactor was
probably homogeneous and phenol was dissociated to a certain
extent as discussed previously. In order to determine the effect
of phase behavior and the dissociation of phenol, we conducted
the experiment at various water densities from 0 g cm23 (in Ar
atmosphere) to 0.5 g cm23 and at reaction times of 60 minutes.
The sum of the yield of alkylphenol was less than 2.4% below
0.3 g cm23 of water density. Table 1 shows the yield of propan-
2-ol (1), 2-isopropylphenol (3), 2-propylphenol (4) and
2,6-diisopropylphenol (5) at 673 K and more than 0.3 g cm23 of
water density. The yields of all alkylphenols increased with
increasing water density at more than 0.4 g cm23 of water
density, compared with the yield of propan-2-ol. This result
clearly shows that alkylation was accelerated with increasing
Chem. Commun., 2001, 1566–1567
1567

 Sato
Sato
 Sekiguchi
Sekiguchi
 Adschiri
Adschiri
 Arai
Arai