
Journal of Chemical Thermodynamics p. 1283 - 1296 (1999)
Update date:2022-08-10
Topics:
 Eli, Wumanjiang
Eli, Wumanjiang
 Chen, Wenhai
Chen, Wenhai
 Xue, Qunji
Xue, Qunji
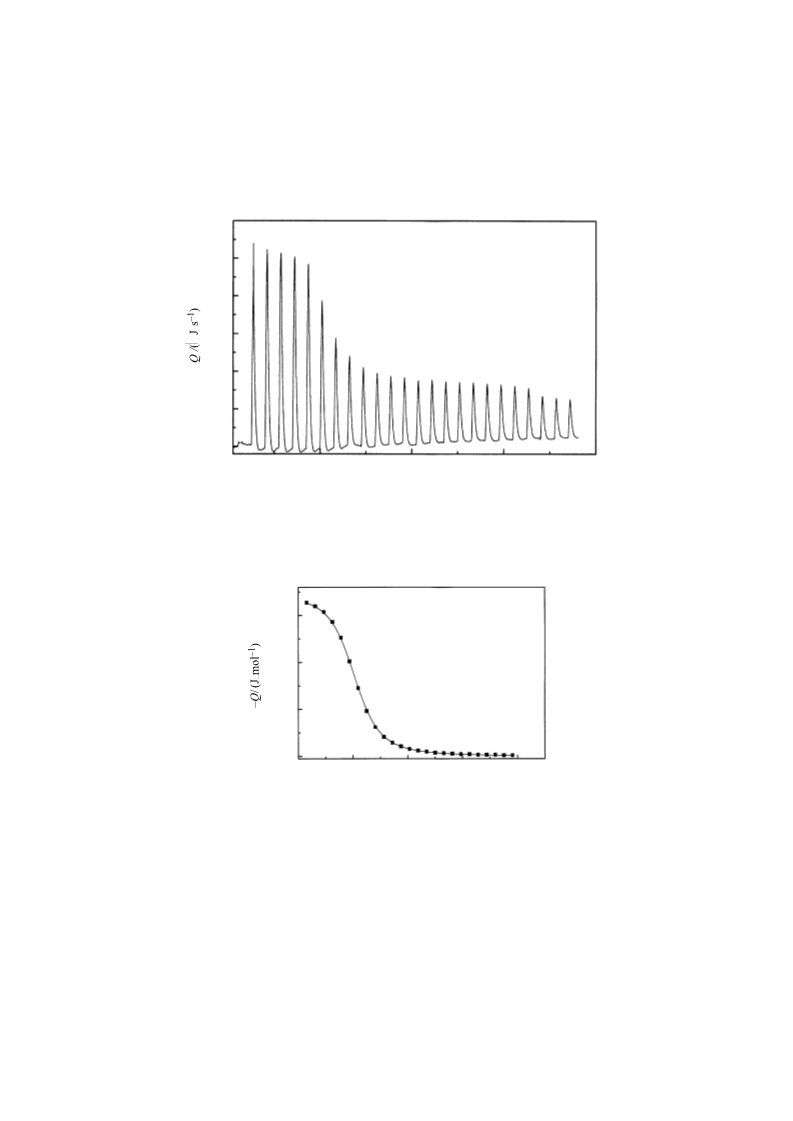
The association of a series of anionic surfactants (CnH2n+1SO4Na, n = 6, 8, 10, 12, 14) with β-cyclodextrin was studied by means of the isothermal titration calorimeter (i.t.c.) at T = 298.15 K. For these types of inclusion complexes, the results agreed well with a 1:1 association mode. Apparent values for the association constants, and changes in the standard molar Gibbs energies, enthalpies, and entropies were derived for the association process. The results indicated that the association of surfactants with β-cyclodextrin is characterized by both favourable enthalpy and favourable entropy changes. The results also demonstrated that the longer the alkyl chain of the anionic surfactant, the greater the association constant with β-cyclodextrin.
View More









Contact:0792-8228321
Address:10TH Floor No.121 binjiang Road Xunyang District
Jiangsu Zhenfang Chemical CO.,LTD.(Suzhou Zhenfang Chemical Factory)
Contact:+86-512-69598882
Address:Room1201, Jiayuan Road No.1018, Xiangcheng District, Suzhou, China
Anhui Sholon New Material Technology Co., Ltd.
website:http://www.sholonchem.com
Contact:+86-550-5261666
Address:4/F Block B, Beijing Chemical Building.No.520 Tianrun Road ,Science & Education Town Wujin District, Changzhou City Jiangsu Province
Improve Medical Technology(Nanxiong) Co., Ltd
Contact:86-751-3836997
Address:No.33, Pingan First Road, Fine Chemical Industry Base, Nanxiong City, Shaoguan, Guangdong, China
Anhui Xinyuan Technology Co.,Ltd
website:http://www.ahxytech.com/
Contact:0086-559-3515800
Address:Huangshan City of Anhui Province Huizhou Huizhou District Road, 21-9
Doi:10.1021/ja01374a036
(1930)Doi:10.1271/bbb.63.238
(1999)Doi:10.1021/ol991151d
(1999)Doi:10.1016/j.jpcs.2009.11.012
(2010)Doi:10.1039/b605091c
(2006)Doi:10.1016/j.molstruc.2019.127269
(2020)