Published on Web 11/06/2007
Widely Useful DMAP-Catalyzed Esterification under Auxiliary
Base- and Solvent-Free Conditions
Akira Sakakura, Kimio Kawajiri, Takuro Ohkubo, Yuji Kosugi, and Kazuaki Ishihara*
Contribution from the Graduate School of Engineering, Nagoya UniVersity, Furo-cho, Chikusa,
Nagoya 464-8603, Japan
Received August 3, 2007; E-mail: ishihara@cc.nagoya-u.ac.jp
Abstract: With regard to atom economy and E-factor, catalytic condensation of carboxylic acids with
equimolar amounts of alcohols is the most desirable. Although several highly active dehydration catalysts
have been reported, more efficient alternatives are still strongly needed because the dehydrative esterification
of tertiary alcohols, phenols, acid-sensitive alcohols, amino acids, and hardly soluble alcohols has never
proceeded satisfactorily. Here we report new insights into the classical DMAP-catalyzed acylation of
alcohols: surprisingly, only a 0.05-2 mol % of DMAP can efficiently promote acylation of alcohols with
acid anhydrides under auxiliary base- and solvent-free conditions to give the corresponding esters in high
yields. Furthermore, we achieved the recovery and reuse of commercially available polystyrene-supported
DMAP without using any solvents. These serendipitous findings provide widely useful and environmentally
benign esterification methods, which might be more practical and reliable than catalytic dehydrative
condensation methods, in particular, for the less reactive alcohols which hardly condense with carboxylic
acid directly.
Introduction
acylation with acid anhydrides is advantageous for the acylation
of less reactive alcohols since the reactivity of acid anhydrides
Catalytic dehydrative condensations between equimolar
amounts of carboxylic acids and alcohols have been developed
as atom-economically ideal synthetic methods of esters over
the decade.1 However, these methods are problematic for the
esterification of sterically demanding tertiary alcohols, less
nucleophilic phenols, acid-sensitive allyl alcohols, amino alco-
hols, and alcohols which are hardly soluble in less polar solvents.
Therefore, more efficient alternatives are still in strong demand.
4-(N,N-Dimethylamino)pyridine (DMAP, 1) is a very effective
nucleophilic base catalyst for the esterification of alcohols with
acid anhydrides2-4 and other related reactions.5,6 The 1-catalyzed
is much higher than that of carboxylic acids. However, it had
been strongly believed for a long period that the classical
1-catalyzed acylation of alcohols should be inferior to the
dehydrative condensation with regard to atom economy and
E-factor, because more than 1 equiv of an auxiliary base would
be required as a scavenger of the carboxylic acid. In contrast,
although Lewis acids such as Sc(OTf)3,7 MoO2Cl2,8 and Bi-
9
(OTf)3 can catalyze the acylation of alcohols with acid
anhydrides without an auxiliary base, the substrate scope is
limited because of strongly acidic conditions.
Here we report new insights into the classical 1-catalyzed
acylation of alcohols: surprisingly, only 0.05-2 mol % of 1
can efficiently promote acylation of alcohols (up to 100 mmol
scale) with equimolar amounts of acid anhydrides under
auxiliary base- and solvent-free conditions to give the corre-
sponding esters quantitatively. Notably, distillable esters can
be synthesized without using solvents throughout the reaction
process including purification.
(1) (a) Otera, J. Esterification; Wiley-VCH: Weinheim, Germany, 2003. (b)
Ishihara, K.; Ohara, S.; Yamamoto, H. Science 2000, 290, 1140. (c) Ishihara,
K.; Nakayama, M.; Ohara, S.; Yamamoto, H. Tetrahedron 2002, 58, 8179.
(d) Ishihara, K.; Nakagawa, S.; Sakakura, A. J. Am. Chem. Soc. 2005, 127,
4168. (e) Sakakura, A.; Nakagawa, S.; Ishihara, K. Tetrahedron 2006, 62,
422. (f) Sakakura, A.; Nakagawa, S.; Ishihara, K. Nat. Protoc. 2007, 2,
1746. (g) Wakasugi, K.; Misaki, T.; Yamada, K.; Tanabe, Y. Tetrahedron
Lett. 2000, 41, 5249. (h) Funatomi, T.; Wakasugi, K.; Misaki, T.; Tanabe,
Y. Green Chem. 2006, 8, 1022. (i) Manabe, K.; Sun, X.-M.; Kobayashi, S.
J. Am. Chem. Soc. 2001, 123, 10101. (j) Xiang, J.; Toyoshima, S.; Orita,
A.; Otera, J. Angew. Chem., Int. Ed. 2001, 40, 3670.
(2) (a) Litivinenko, L. M.; Kirichenko, A. I. Dokl. Akad. Nauk. SSSR 1967,
176, 97. (b) Steglich, W.; Ho¨fle, G. Angew. Chem., Int. Ed. 1969, 8, 981.
(c) Ho¨fle, G.; Steglich, W. Synthesis 1972, 619.
Results and Discussion
(3) Reviews: (a) Ho¨fle, G.; Steglich, W.; Vorbru¨ggen, H. Angew. Chem., Int.
Ed. 1978, 17, 569. (b) Scriven, E. F. V. Chem. Soc. ReV. 1983, 12, 129.
(c) Ragnarsson, U.; Grehn, L. Acc. Chem. Res. 1998, 31, 494. (d) Grondal,
C. Synlett 2003, 1568. (e) Spivey, A. C.; Arseniyadis, S. Angew. Chem.,
Int. Ed. 2004, 43, 5436.
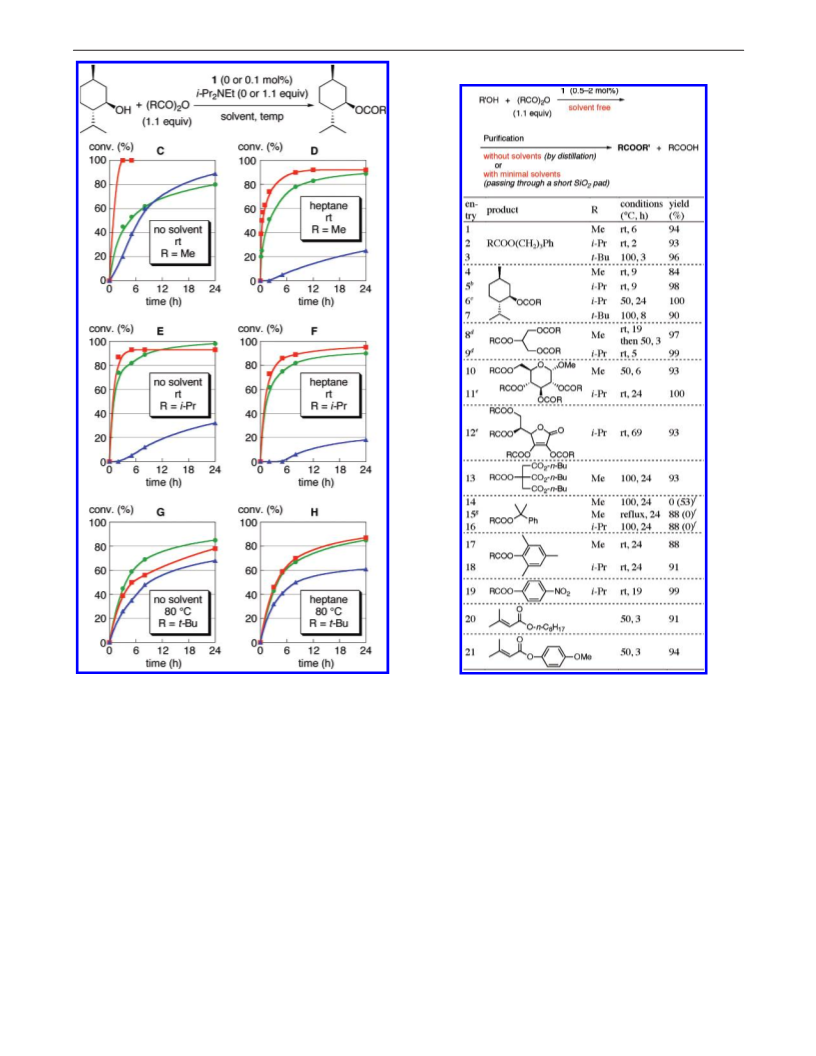
Effects of Solvent and Auxiliary Base on the DMAP-
Catalyzed Acylations. Recently, Zipse and co-workers proposed
a mechanism for the 1-catalyzed acetylation of alcohols (Figure
(4) Tributylphosphine is also known as nucleophilic catalyst for acylation. (a)
Vedejs, E.; Diver, S. T. J. Am. Chem. Soc. 1993, 115, 3358. (b) Vedejs,
E.; Bennett, N. S.; Conn, L. M.; Diver, S. T.; Gingras, M.; Lin, S.; Oliver,
P. A.; Peterson, M. J. J. Org. Chem. 1993, 58, 7286.
(5) Macrolactonization: (a) Inanaga, J.; Hirata, K.; Saeki, H.; Katsuki, T.;
Yamaguchi, M. Bull. Chem. Soc. Jpn. 1979, 52, 1989. (b) Boden, E. P.;
Keck, G. E. J. Org. Chem. 1985, 50, 2394.
(6) Silylation: Chaudhary, S. K.; Hernandez, O. Tetrahedron Lett. 1979, 20,
99.
(7) (a) Ishihara, K.; Kubota, M.; Kurihara, H.; Yamamoto, H. J. Am. Chem.
Soc. 1995, 117, 4413. (b) Ishihara, K.; Kubota, M.; Kurihara, H.;
Yamamoto, H. J. Org. Chem. 1996, 61, 4560. (c) Ishihara, K.; Kubota,
M.; Yamamoto, H. Synlett 1996, 265.
(8) Chen, C.-T.; Kuo, J.-H.; Pawar, V. D.; Munot, Y. S.; Weng, S.-S.; Ku,
C.-H.; Liu, C.-Y. J. Org. Chem. 2005, 70, 1188.
(9) Orita, A.; Tanahashi, C.; Kakuda, A.; Otera, J. J. Org. Chem. 2001, 66,
8926.
9
10.1021/ja075824w CCC: $37.00 © 2007 American Chemical Society
J. AM. CHEM. SOC. 2007, 129, 14775-14779
14775

 Sakakura, Akira
Sakakura, Akira
 Kawajiri, Kimio
Kawajiri, Kimio
 Ohkubo, Takuro
Ohkubo, Takuro
 Kosugi, Yuji
Kosugi, Yuji
 Ishihara, Kazuaki
Ishihara, Kazuaki