
Journal of Solution Chemistry p. 775 - 784 (1981)
Update date:2022-08-11
Topics:
 Hoeiland, H.
Hoeiland, H.
 Hald, L.H.
Hald, L.H.
 Kvammen, O.J.
Kvammen, O.J.
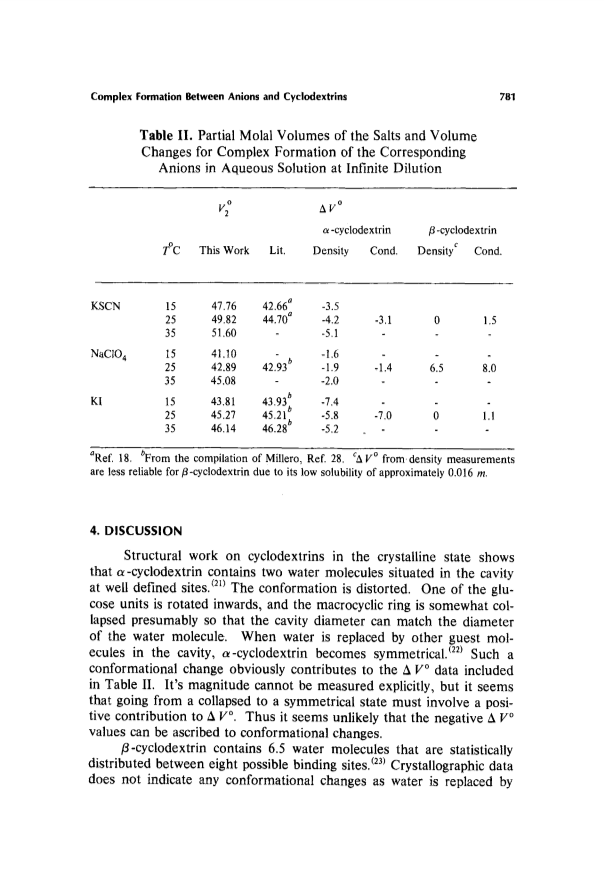
Partial molal volume changes during complex formation between SCN(1-), I(1-), and ClO4(1-) and α- and β-cyclodextrin have been determined by two independent methods of measurements; one based on density measurement and subsequent calculation of apparent molal volumes, the other on differentiating the association constants with respect to pressure.Results from the two methods are in good agreement.Negative volume changes were observed for complex formation between the anions and α-cyclodextrin while zero or slightly positive values were observed for complex formation with β-cyclodextrin.The result is consistent with the idea that the anions do not become dehydrated as they form complexes with cyclodextrins.
View More








Dongguan Albiya Energy Science and Technology Co.,Ltd
Contact:+86-769-22181286
Address:Huanan Industial Park, Dongguan,China
Contact:+86-371-55981030
Address:Room 1571, Macalline Soho, No.1, Shangdu Road, Zhengzhou, Henan
Ceresking Ecology & Technology co.,ltd
Contact:86 22 66218397
Address:Room 1613, Zheshang Mansion, No. 1988, Yingbin Avenue, Binhai New District, Tianjin,China.
website:http://www.sagechem.com
Contact:+86-571-86818502
Address:Room C1301, New Youth Plaza, 8 Jia Shan Road, Hangzhou, China
shanghai jiuling chemical co.,ltd.
Contact:+86-21-50387295
Address:Zaozhuang Road, Pudong, Shanghai City. China
Doi:10.1016/j.tetasy.2009.07.048
(2009)Doi:10.1016/j.molcata.2015.05.009
(2015)Doi:10.1021/jo01362a028
(1957)Doi:10.1039/j29710001625
(1971)Doi:10.1021/ja01245a513
(1943)Doi:10.1021/jm00333a045
(1964)