
Canadian Journal of Chemistry p. 3061 - 3070 (1982)
Update date:2022-08-24
Topics:
 Cox, Robin A.
Cox, Robin A.
 Yates, Keith
Yates, Keith
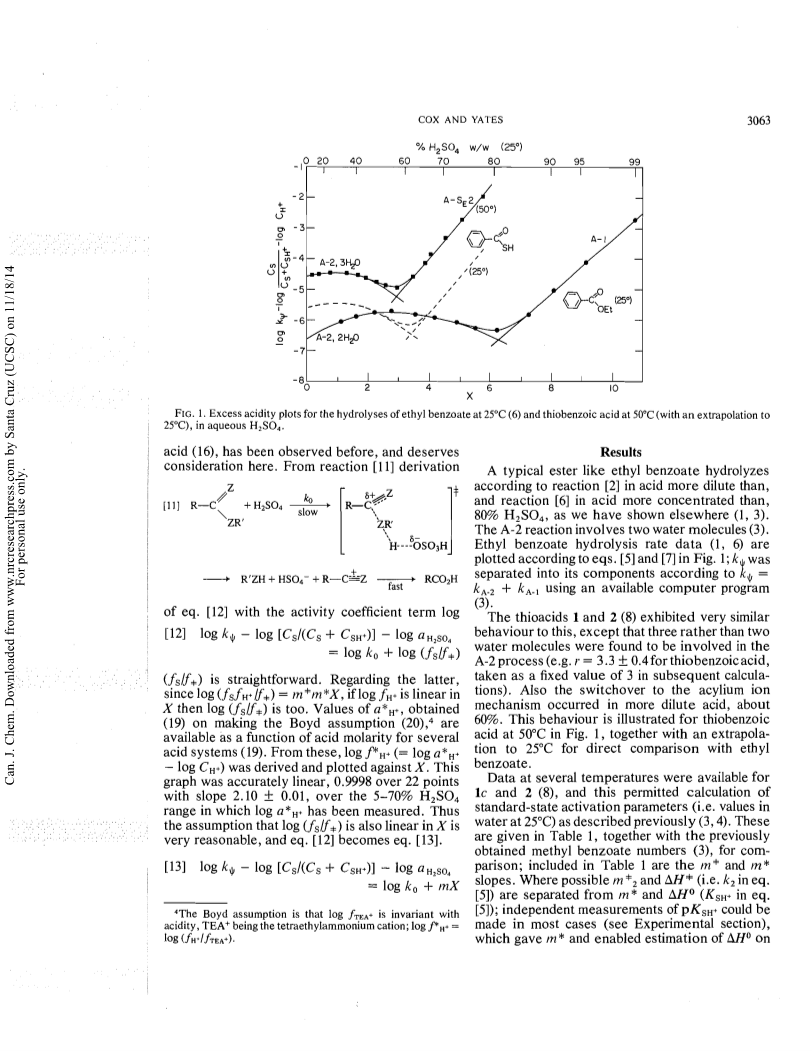
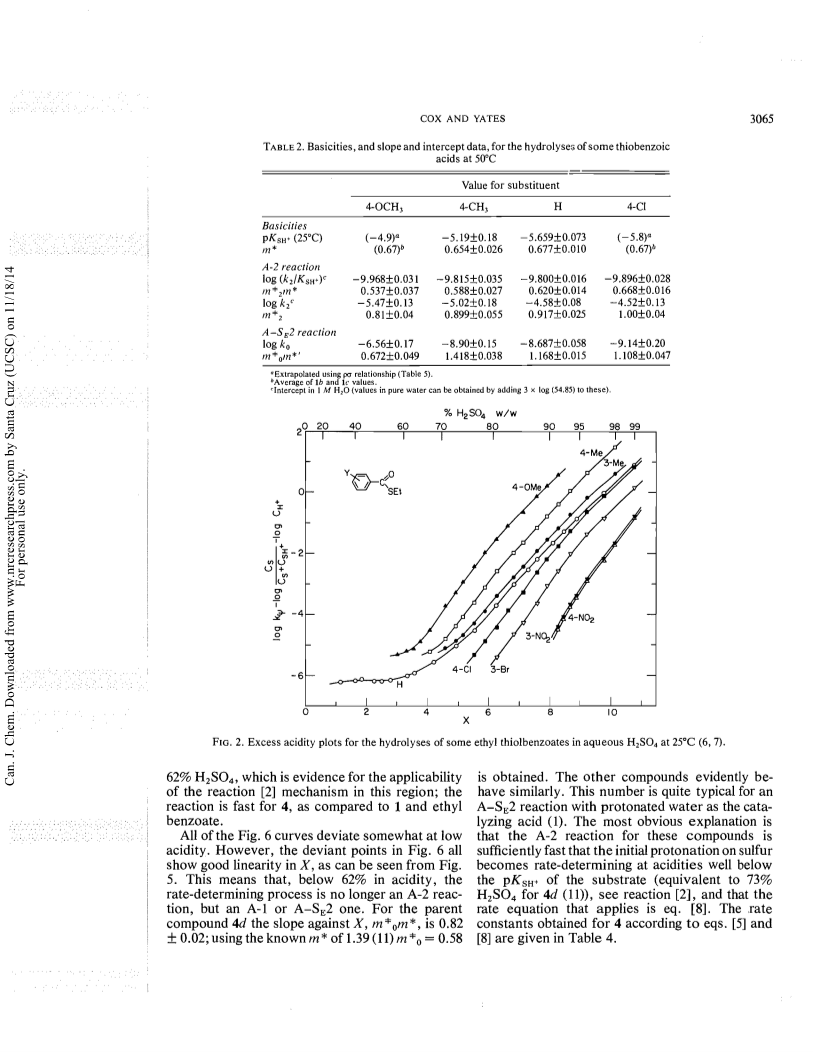
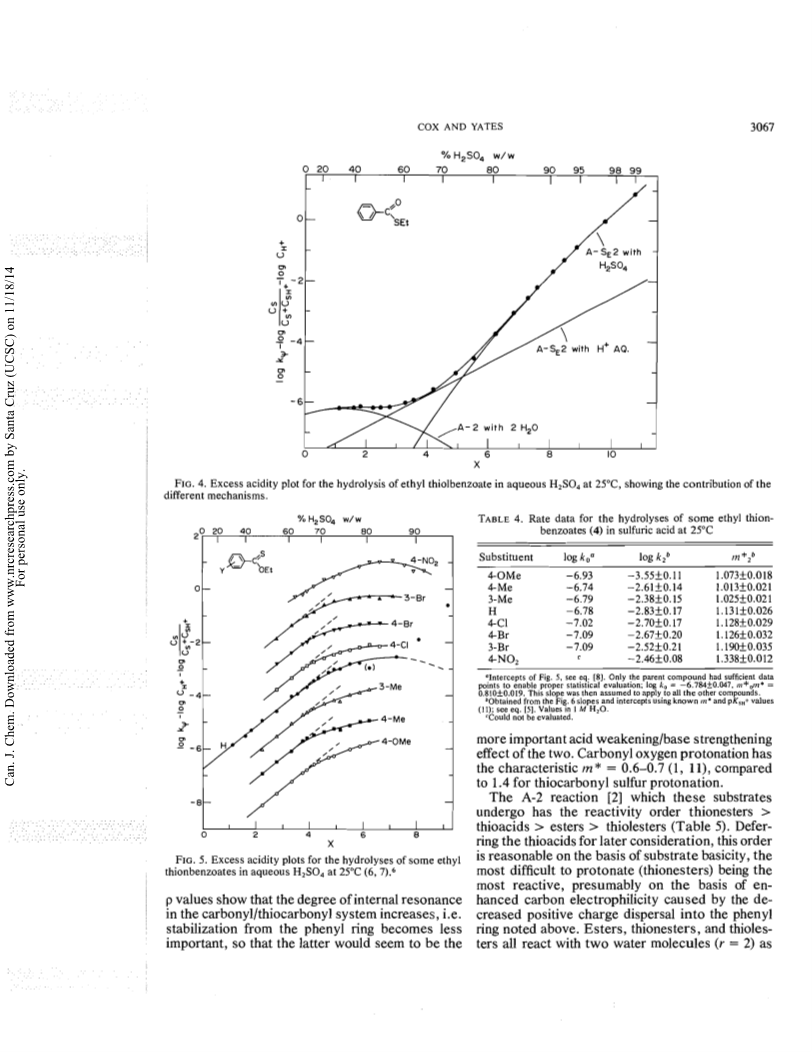
The excess acidity method has been applied to hydrolysis rate data, obtained as a function of medium composition, for four thiobenzoic acids, thioacetic acid, eight ethyl thiolbenzoates, and eight ethyl thionbenzoates in aqueous sulfuric acid.The mechanistic behaviour thus revealed has both similarities to and differences from that of a typical ester like ethyl benzoate, which gives benzoic acid by an A-2 reaction involving two water molecules in weak acid, and by A-1 acylium ion formation in strong acid.The thioacids follow this behaviour, except that the A-2process involves three water molecules, and that the mechanistic changeover occurs in 60percent rather than 80percent acid.The A-2 process for the ethyl thiolbenzoates is slow; the major hydrolysis mechanism is acylium ion formation, not in an A-1 reaction but by a concerted A-SE2 process involving both proton transfer to sulfur and carbon-sulfur bond breaking.The major proton transfer agent is the undissociated sulfuric acid molecule.The thionbenzoate esters, in contrast, undergo very fast A-2 hydrolysis; so fast, in fact, that the initial protonation of sulfur is the rate-determining step in acids more dilute than about 62percent w/w.It appears that proton transfer to sulfur is a comparatively slow process.
View More






SHENZHEN PENGCHENG REDSTAR INDUSTRY CO.,LTD
Contact:+86-755-82412922
Address:Room 8066, East Block, Square City, Jiabin Road, Luohu District
Feis International Trade Co,. Ltd
Contact:13961823444-18235944442
Address:Wuxi jiangsu
Contact:+86-574-89075960
Address:#100 Xiang Yun Road, New High tech area, Ningbo, China
Contact:86-15588110016
Address:LINYI CITY,SHANDONG PROVINCE,CHINA
Contact:86 513 85512619
Address:Rm.1306, Building A, Wenfeng Mansion,168 Gongnong Road, Nantong Jiangsu China
Doi:10.1039/c4cc06390b
(2014)Doi:10.1007/BF00750880
(1982)Doi:10.1080/00397919708007297
(1997)Doi:10.1021/ol052517r
(2006)Doi:10.1016/0040-4020(95)00795-A
(1995)Doi:10.1016/j.tet.2003.08.027
(2003)